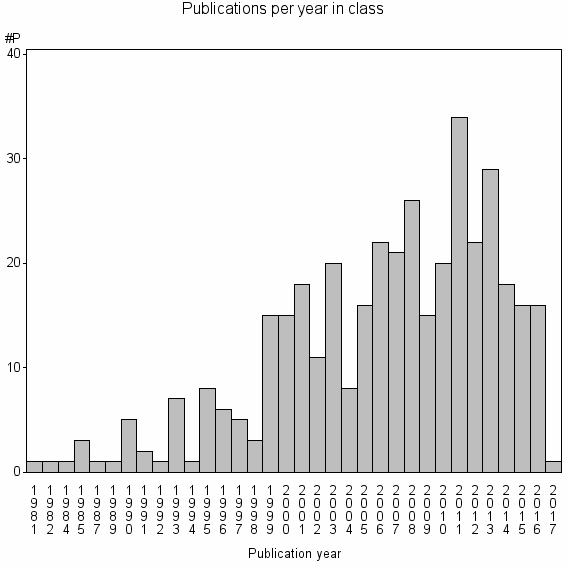
Class information for: |
Basic class information |
| Class id | #P | Avg. number of references |
Database coverage of references |
|---|---|---|---|
| 22251 | 389 | 22.6 | 58% |
Hierarchy of classes |
The table includes all classes above and classes immediately below the current class. |
| Cluster id | Level | Cluster label | #P |
|---|---|---|---|
| 9 | 4 | COMPUTER SCIENCE, THEORY & METHODS//COMPUTER SCIENCE, ARTIFICIAL INTELLIGENCE//COMPUTER SCIENCE, INFORMATION SYSTEMS | 1247339 |
| 34 | 3 | STATISTICS & PROBABILITY//STAT//COMMUNICATIONS IN STATISTICS-THEORY AND METHODS | 105938 |
| 1075 | 2 | ADAPTIVE DESIGN//STATISTICS IN MEDICINE//STATISTICS & PROBABILITY | 9928 |
| 22251 | 1 | NONINFERIORITY MARGIN//NON INFERIORITY//THREE ARM DESIGN | 389 |
Terms with highest relevance score |
| rank | Term | termType | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|---|
| 1 | NONINFERIORITY MARGIN | authKW | 1577960 | 7% | 72% | 28 |
| 2 | NON INFERIORITY | authKW | 756588 | 11% | 23% | 42 |
| 3 | THREE ARM DESIGN | authKW | 706466 | 2% | 100% | 9 |
| 4 | ACTIVE CONTROL TRIALS | authKW | 678427 | 3% | 79% | 11 |
| 5 | NON INFERIORITY TRIAL | authKW | 668886 | 7% | 30% | 28 |
| 6 | NONINFERIORITY | authKW | 488378 | 7% | 22% | 28 |
| 7 | FIXED MARGIN METHOD | authKW | 470978 | 2% | 100% | 6 |
| 8 | CONSTANCY ASSUMPTION | authKW | 403693 | 2% | 86% | 6 |
| 9 | BIO CREEP | authKW | 392481 | 1% | 100% | 5 |
| 10 | FIXED MARGIN APPROACH | authKW | 392481 | 1% | 100% | 5 |
Web of Science journal categories |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | Statistics & Probability | 10280 | 40% | 0% | 154 |
| 2 | Medical Informatics | 7306 | 17% | 0% | 66 |
| 3 | Mathematical & Computational Biology | 6990 | 25% | 0% | 96 |
| 4 | Health Care Sciences & Services | 1224 | 12% | 0% | 48 |
| 5 | Pharmacology & Pharmacy | 878 | 29% | 0% | 113 |
| 6 | Public, Environmental & Occupational Health | 722 | 19% | 0% | 72 |
| 7 | Medicine, Research & Experimental | 708 | 17% | 0% | 65 |
| 8 | Medicine, General & Internal | 158 | 11% | 0% | 42 |
| 9 | Ethics | 72 | 2% | 0% | 6 |
| 10 | MEDICINE, MISCELLANEOUS | 48 | 0% | 0% | 1 |
Address terms |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | OBOPASS | 156993 | 1% | 100% | 2 |
| 2 | BIOMETR 4 | 135266 | 2% | 19% | 9 |
| 3 | BIOMETR 1 | 128666 | 3% | 16% | 10 |
| 4 | OPASS | 117741 | 1% | 50% | 3 |
| 5 | PHARMACEUT LOGIST | 104660 | 1% | 67% | 2 |
| 6 | BIOMETR 6 | 91147 | 2% | 19% | 6 |
| 7 | ABLEH | 78496 | 0% | 100% | 1 |
| 8 | ABT KLIN PHARMAKOKINET | 78496 | 0% | 100% | 1 |
| 9 | BIOSTAT MED WRITING UK | 78496 | 0% | 100% | 1 |
| 10 | CDEROFF TRANSLAT SCI | 78496 | 0% | 100% | 1 |
Journals |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | PHARMACEUTICAL STATISTICS | 72324 | 6% | 4% | 22 |
| 2 | JOURNAL OF BIOPHARMACEUTICAL STATISTICS | 72171 | 7% | 3% | 29 |
| 3 | STATISTICS IN BIOPHARMACEUTICAL RESEARCH | 50417 | 4% | 5% | 14 |
| 4 | DRUG INFORMATION JOURNAL | 44673 | 6% | 2% | 25 |
| 5 | STATISTICS IN MEDICINE | 33157 | 14% | 1% | 55 |
| 6 | BIOMETRICAL JOURNAL | 7435 | 4% | 1% | 15 |
| 7 | CONTROLLED CLINICAL TRIALS | 6537 | 2% | 1% | 9 |
| 8 | STATISTICAL METHODS IN MEDICAL RESEARCH | 5205 | 2% | 1% | 7 |
| 9 | CLINICAL TRIALS | 3797 | 2% | 1% | 6 |
| 10 | SCIENCE AND ENGINEERING ETHICS | 1332 | 1% | 0% | 4 |
Author Key Words |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
LCSH search | Wikipedia search |
|---|---|---|---|---|---|---|---|
| 1 | NONINFERIORITY MARGIN | 1577960 | 7% | 72% | 28 | Search NONINFERIORITY+MARGIN | Search NONINFERIORITY+MARGIN |
| 2 | NON INFERIORITY | 756588 | 11% | 23% | 42 | Search NON+INFERIORITY | Search NON+INFERIORITY |
| 3 | THREE ARM DESIGN | 706466 | 2% | 100% | 9 | Search THREE+ARM+DESIGN | Search THREE+ARM+DESIGN |
| 4 | ACTIVE CONTROL TRIALS | 678427 | 3% | 79% | 11 | Search ACTIVE+CONTROL+TRIALS | Search ACTIVE+CONTROL+TRIALS |
| 5 | NON INFERIORITY TRIAL | 668886 | 7% | 30% | 28 | Search NON+INFERIORITY+TRIAL | Search NON+INFERIORITY+TRIAL |
| 6 | NONINFERIORITY | 488378 | 7% | 22% | 28 | Search NONINFERIORITY | Search NONINFERIORITY |
| 7 | FIXED MARGIN METHOD | 470978 | 2% | 100% | 6 | Search FIXED+MARGIN+METHOD | Search FIXED+MARGIN+METHOD |
| 8 | CONSTANCY ASSUMPTION | 403693 | 2% | 86% | 6 | Search CONSTANCY+ASSUMPTION | Search CONSTANCY+ASSUMPTION |
| 9 | BIO CREEP | 392481 | 1% | 100% | 5 | Search BIO+CREEP | Search BIO+CREEP |
| 10 | FIXED MARGIN APPROACH | 392481 | 1% | 100% | 5 | Search FIXED+MARGIN+APPROACH | Search FIXED+MARGIN+APPROACH |
Core articles |
The table includes core articles in the class. The following variables is taken into account for the relevance score of an article in a cluster c: (1) Number of references referring to publications in the class. (2) Share of total number of active references referring to publications in the class. (3) Age of the article. New articles get higher score than old articles. (4) Citation rate, normalized to year. |
| Rank | Reference | # ref. in cl. |
Shr. of ref. in cl. |
Citations |
|---|---|---|---|---|
| 1 | HUNG, HMJ , WANG, SJ , (2013) STATISTICAL CONSIDERATIONS FOR NONINFERIORITY TRIAL DESIGNS WITHOUT PLACEBO.STATISTICS IN BIOPHARMACEUTICAL RESEARCH. VOL. 5. ISSUE 3. P. 239 -247 | 27 | 84% | 2 |
| 2 | HUNG, HMJ , WANG, SJ , O'NEILL, R , (2009) CHALLENGES AND REGULATORY EXPERIENCES WITH NON-INFERIORITY TRIAL DESIGN WITHOUT PLACEBO ARM.BIOMETRICAL JOURNAL. VOL. 51. ISSUE 2. P. 324-334 | 24 | 96% | 20 |
| 3 | SOON, GX , ZHANG, ZW , TSONG, Y , NIE, L , (2013) ASSESSING OVERALL EVIDENCE FROM NONINFERIORITY TRIALS WITH SHARED HISTORICAL DATA.STATISTICS IN MEDICINE. VOL. 32. ISSUE 14. P. 2349-2363 | 22 | 88% | 1 |
| 4 | HUNG, HMJ , WANG, SJ , O'NEILL, R , (2007) ISSUES WITH STATISTICAL RISKS FOR TESTING METHODS IN NONINFERIORITY TRIAL WITHOUT A PLACEBO ARM.JOURNAL OF BIOPHARMACEUTICAL STATISTICS. VOL. 17. ISSUE 2. P. 201-213 | 24 | 89% | 12 |
| 5 | KANG, SH , (2016) AVOIDING AMBIGUITY WITH THE TYPE I ERROR RATE IN NONINFERIORITY TRIALS.JOURNAL OF BIOPHARMACEUTICAL STATISTICS. VOL. 26. ISSUE 3. P. 452 -465 | 22 | 76% | 0 |
| 6 | NIE, L , SOON, G , (2010) A COVARIATE-ADJUSTMENT REGRESSION MODEL APPROACH TO NONINFERIORITY MARGIN DEFINITION.STATISTICS IN MEDICINE. VOL. 29. ISSUE 10. P. 1107 -1113 | 17 | 100% | 3 |
| 7 | GUPTA, SK , (2011) NON-INFERIORITY CLINICAL TRIALS: PRACTICAL ISSUES AND CURRENT REGULATORY PERSPECTIVE.INDIAN JOURNAL OF PHARMACOLOGY. VOL. 43. ISSUE 4. P. 371-374 | 16 | 100% | 4 |
| 8 | XU, SY , BARKER, K , MENON, S , D'AGOSTINO, RB , (2014) COVARIATE EFFECT ON CONSTANCY ASSUMPTION IN NONINFERIORITY CLINICAL TRIALS.JOURNAL OF BIOPHARMACEUTICAL STATISTICS. VOL. 24. ISSUE 6. P. 1173 -1189 | 16 | 89% | 0 |
| 9 | HUNG, HMJ , WANG, SJ , O'NEILL, R , (2005) A REGULATORY PERSPECTIVE ON CHOICE OF MARGIN AND STATISTICAL INFERENCE ISSUE IN NON-INFERIORITY TRIALS.BIOMETRICAL JOURNAL. VOL. 47. ISSUE 1. P. 28-36 | 19 | 83% | 60 |
| 10 | SNAPINN, S , JIANG, Q , (2008) PRESERVATION OF EFFECT AND THE REGULATORY APPROVAL OF NEW TREATMENTS ON THE BASIS OF NON-INFERIORITY TRIALS.STATISTICS IN MEDICINE. VOL. 27. ISSUE 3. P. 382-391 | 15 | 100% | 14 |
Classes with closest relation at Level 1 |