Class information for: |
Basic class information |
| Class id | #P | Avg. number of references |
Database coverage of references |
|---|---|---|---|
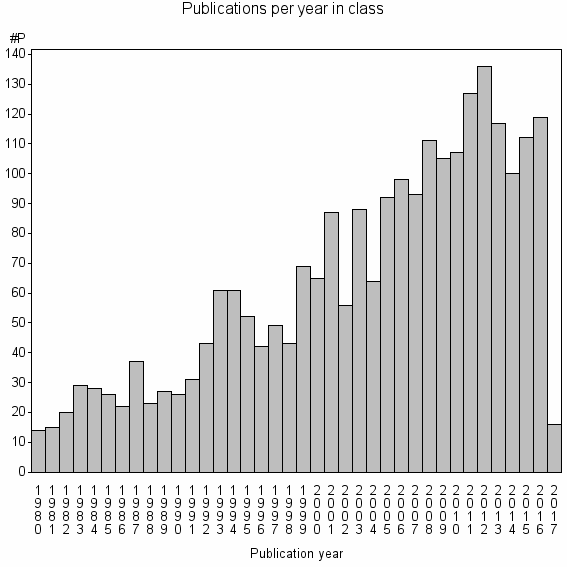
| 1949 | 2411 | 22.9 | 61% |
Hierarchy of classes |
The table includes all classes above and classes immediately below the current class. |
| Cluster id | Level | Cluster label | #P |
|---|---|---|---|
| 9 | 4 | COMPUTER SCIENCE, THEORY & METHODS//COMPUTER SCIENCE, ARTIFICIAL INTELLIGENCE//COMPUTER SCIENCE, INFORMATION SYSTEMS | 1247339 |
| 34 | 3 | STATISTICS & PROBABILITY//STAT//COMMUNICATIONS IN STATISTICS-THEORY AND METHODS | 105938 |
| 1075 | 2 | ADAPTIVE DESIGN//STATISTICS IN MEDICINE//STATISTICS & PROBABILITY | 9928 |
| 1949 | 1 | ADAPTIVE DESIGN//INTERIM ANALYSIS//GROUP SEQUENTIAL TEST | 2411 |
Terms with highest relevance score |
| rank | Term | termType | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|---|
| 1 | ADAPTIVE DESIGN | authKW | 1533562 | 10% | 48% | 250 |
| 2 | INTERIM ANALYSIS | authKW | 1285928 | 6% | 73% | 140 |
| 3 | GROUP SEQUENTIAL TEST | authKW | 918953 | 3% | 92% | 79 |
| 4 | CONDITIONAL POWER | authKW | 911727 | 3% | 86% | 84 |
| 5 | SAMPLE SIZE RE ESTIMATION | authKW | 900477 | 3% | 89% | 80 |
| 6 | GROUP SEQUENTIAL DESIGN | authKW | 840731 | 3% | 84% | 79 |
| 7 | INTERIM ANALYSES | authKW | 515285 | 2% | 88% | 46 |
| 8 | STATISTICS IN MEDICINE | journal | 399385 | 20% | 7% | 475 |
| 9 | TWO STAGE DESIGN | authKW | 388387 | 3% | 46% | 66 |
| 10 | DATA MONITORING COMMITTEE | authKW | 370379 | 2% | 75% | 39 |
Web of Science journal categories |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | Statistics & Probability | 154171 | 61% | 1% | 1477 |
| 2 | Mathematical & Computational Biology | 96472 | 37% | 1% | 884 |
| 3 | Medical Informatics | 85890 | 23% | 1% | 562 |
| 4 | Medicine, Research & Experimental | 12683 | 27% | 0% | 663 |
| 5 | Public, Environmental & Occupational Health | 7005 | 23% | 0% | 549 |
| 6 | Pharmacology & Pharmacy | 3446 | 24% | 0% | 572 |
| 7 | Biology | 2399 | 9% | 0% | 229 |
| 8 | Health Care Sciences & Services | 1892 | 6% | 0% | 154 |
| 9 | Oncology | 181 | 7% | 0% | 161 |
| 10 | Computer Science, Interdisciplinary Applications | 50 | 2% | 0% | 46 |
Address terms |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | MED PHARMACEUT STAT UNIT | 269555 | 2% | 41% | 52 |
| 2 | MED BIOMETRY UNIT | 109442 | 0% | 79% | 11 |
| 3 | BIOMETR 1 | 91521 | 1% | 34% | 21 |
| 4 | OB CDER | 37990 | 0% | 100% | 3 |
| 5 | COMPETENCE CLIN TRIALS | 35171 | 0% | 56% | 5 |
| 6 | BIOMETRY MATH STAT BRANCH | 32904 | 1% | 20% | 13 |
| 7 | STAT DRUG DEV | 32556 | 0% | 43% | 6 |
| 8 | OTS CDER | 31653 | 0% | 50% | 5 |
| 9 | BIOMETR ADV ANALYT | 25326 | 0% | 100% | 2 |
| 10 | BIOTX STAT | 25326 | 0% | 100% | 2 |
Journals |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | STATISTICS IN MEDICINE | 399385 | 20% | 7% | 475 |
| 2 | JOURNAL OF BIOPHARMACEUTICAL STATISTICS | 279127 | 6% | 16% | 142 |
| 3 | PHARMACEUTICAL STATISTICS | 222137 | 4% | 18% | 96 |
| 4 | CONTROLLED CLINICAL TRIALS | 213669 | 5% | 13% | 128 |
| 5 | CLINICAL TRIALS | 114608 | 3% | 11% | 82 |
| 6 | STATISTICS IN BIOPHARMACEUTICAL RESEARCH | 80305 | 2% | 14% | 44 |
| 7 | BIOMETRICS | 79171 | 7% | 4% | 168 |
| 8 | BIOMETRICAL JOURNAL | 46112 | 4% | 4% | 93 |
| 9 | DRUG INFORMATION JOURNAL | 45697 | 3% | 6% | 63 |
| 10 | CONTEMPORARY CLINICAL TRIALS | 31351 | 2% | 4% | 59 |
Author Key Words |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
LCSH search | Wikipedia search |
|---|---|---|---|---|---|---|---|
| 1 | ADAPTIVE DESIGN | 1533562 | 10% | 48% | 250 | Search ADAPTIVE+DESIGN | Search ADAPTIVE+DESIGN |
| 2 | INTERIM ANALYSIS | 1285928 | 6% | 73% | 140 | Search INTERIM+ANALYSIS | Search INTERIM+ANALYSIS |
| 3 | GROUP SEQUENTIAL TEST | 918953 | 3% | 92% | 79 | Search GROUP+SEQUENTIAL+TEST | Search GROUP+SEQUENTIAL+TEST |
| 4 | CONDITIONAL POWER | 911727 | 3% | 86% | 84 | Search CONDITIONAL+POWER | Search CONDITIONAL+POWER |
| 5 | SAMPLE SIZE RE ESTIMATION | 900477 | 3% | 89% | 80 | Search SAMPLE+SIZE+RE+ESTIMATION | Search SAMPLE+SIZE+RE+ESTIMATION |
| 6 | GROUP SEQUENTIAL DESIGN | 840731 | 3% | 84% | 79 | Search GROUP+SEQUENTIAL+DESIGN | Search GROUP+SEQUENTIAL+DESIGN |
| 7 | INTERIM ANALYSES | 515285 | 2% | 88% | 46 | Search INTERIM+ANALYSES | Search INTERIM+ANALYSES |
| 8 | TWO STAGE DESIGN | 388387 | 3% | 46% | 66 | Search TWO+STAGE+DESIGN | Search TWO+STAGE+DESIGN |
| 9 | DATA MONITORING COMMITTEE | 370379 | 2% | 75% | 39 | Search DATA+MONITORING+COMMITTEE | Search DATA+MONITORING+COMMITTEE |
| 10 | GROUP SEQUENTIAL | 323157 | 1% | 73% | 35 | Search GROUP+SEQUENTIAL | Search GROUP+SEQUENTIAL |
Core articles |
The table includes core articles in the class. The following variables is taken into account for the relevance score of an article in a cluster c: (1) Number of references referring to publications in the class. (2) Share of total number of active references referring to publications in the class. (3) Age of the article. New articles get higher score than old articles. (4) Citation rate, normalized to year. |
| Rank | Reference | # ref. in cl. |
Shr. of ref. in cl. |
Citations |
|---|---|---|---|---|
| 1 | BAUER, P , BRETZ, F , DRAGALIN, V , KONIG, F , WASSMER, G , (2016) TWENTY-FIVE YEARS OF CONFIRMATORY ADAPTIVE DESIGNS: OPPORTUNITIES AND PITFALLS.STATISTICS IN MEDICINE. VOL. 35. ISSUE 3. P. 325 -347 | 114 | 86% | 7 |
| 2 | VANDEMEULEBROECKE, M , (2008) GROUP SEQUENTIAL AND ADAPTIVE DESIGNS - A REVIEW OF BASIC CONCEPTS AND POINTS OF DISCUSSION.BIOMETRICAL JOURNAL. VOL. 50. ISSUE 4. P. 541 -557 | 76 | 100% | 12 |
| 3 | TODD, S , (2007) A 25-YEAR REVIEW OF SEQUENTIAL METHODOLOGY IN CLINICAL STUDIES.STATISTICS IN MEDICINE. VOL. 26. ISSUE 2. P. 237-252 | 70 | 83% | 19 |
| 4 | BRETZ, F , KOENIG, F , BRANNATH, W , GLIMM, E , POSCH, M , (2009) ADAPTIVE DESIGNS FOR CONFIRMATORY CLINICAL TRIALS.STATISTICS IN MEDICINE. VOL. 28. ISSUE 8. P. 1181 -1217 | 51 | 91% | 60 |
| 5 | PRITCHETT, YL , MENON, S , MARCHENKO, O , ANTONIJEVIC, Z , MILLER, E , SANCHEZ-KAM, M , MORGAN-BOUNIOL, CC , NGUYEN, H , PRUCKA, WR , (2015) SAMPLE SIZE RE-ESTIMATION DESIGNS IN CONFIRMATORY CLINICAL TRIALS-CURRENT STATE, STATISTICAL CONSIDERATIONS, AND PRACTICAL GUIDANCE.STATISTICS IN BIOPHARMACEUTICAL RESEARCH. VOL. 7. ISSUE 4. P. 309 -321 | 47 | 96% | 0 |
| 6 | GRANT, AM , ALTMAN, DG , BABIKER, AB , CAMPBELL, MK , CLEMENS, FJ , DARBYSHIRE, JH , ELBOURNE, DR , MCLEER, SK , PARMAR, MKB , POCOCK, SJ , ET AL (2005) ISSUES IN DATA MONITORING AND INTERIM ANALYSIS OF TRIALS.HEALTH TECHNOLOGY ASSESSMENT. VOL. 9. ISSUE 7. P. 1-+ | 88 | 58% | 54 |
| 7 | HAMPSON, LV , JENNISON, C , (2013) GROUP SEQUENTIAL TESTS FOR DELAYED RESPONSES.JOURNAL OF THE ROYAL STATISTICAL SOCIETY SERIES B-STATISTICAL METHODOLOGY. VOL. 75. ISSUE 1. P. 3 -39 | 39 | 93% | 10 |
| 8 | DRAGALIN, V , (2006) ADAPTIVE DESIGNS: TERMINOLOGY AND CLASSIFICATION.DRUG INFORMATION JOURNAL. VOL. 40. ISSUE 4. P. 425-435 | 53 | 77% | 62 |
| 9 | JENNISON, C , TURNBULL, BW , (2007) ADAPTIVE SEAMLESS DESIGNS: SELECTION AND PROSPECTIVE TESTING OF HYPOTHESES.JOURNAL OF BIOPHARMACEUTICAL STATISTICS. VOL. 17. ISSUE 6. P. 1135-1161 | 48 | 86% | 24 |
| 10 | WASSMER, G , (2000) BASIC CONCEPTS OF GROUP SEQUENTIAL AND ADAPTIVE GROUP SEQUENTIAL TEST PROCEDURES.STATISTICAL PAPERS. VOL. 41. ISSUE 3. P. 253 -279 | 52 | 100% | 17 |
Classes with closest relation at Level 1 |