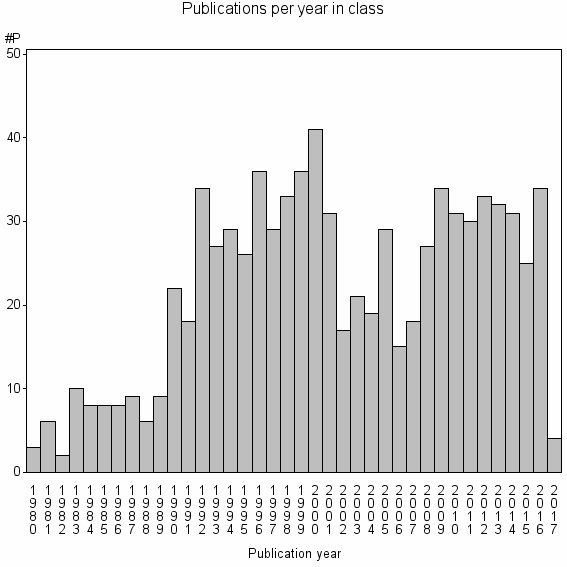
Class information for: |
Basic class information |
| Class id | #P | Avg. number of references |
Database coverage of references |
|---|---|---|---|
| 13520 | 831 | 20.0 | 49% |
Hierarchy of classes |
The table includes all classes above and classes immediately below the current class. |
| Cluster id | Level | Cluster label | #P |
|---|---|---|---|
| 9 | 4 | COMPUTER SCIENCE, THEORY & METHODS//COMPUTER SCIENCE, ARTIFICIAL INTELLIGENCE//COMPUTER SCIENCE, INFORMATION SYSTEMS | 1247339 |
| 34 | 3 | STATISTICS & PROBABILITY//STAT//COMMUNICATIONS IN STATISTICS-THEORY AND METHODS | 105938 |
| 1075 | 2 | ADAPTIVE DESIGN//STATISTICS IN MEDICINE//STATISTICS & PROBABILITY | 9928 |
| 13520 | 1 | BIOEQUIVALENCE//INDIVIDUAL BIOEQUIVALENCE//HIGHLY VARIABLE DRUGS | 831 |
Terms with highest relevance score |
| rank | Term | termType | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|---|
| 1 | BIOEQUIVALENCE | authKW | 1565295 | 32% | 16% | 265 |
| 2 | INDIVIDUAL BIOEQUIVALENCE | authKW | 1081455 | 4% | 89% | 33 |
| 3 | HIGHLY VARIABLE DRUGS | authKW | 900218 | 3% | 88% | 28 |
| 4 | AVERAGE BIOEQUIVALENCE | authKW | 683995 | 3% | 85% | 22 |
| 5 | POPULATION BIOEQUIVALENCE | authKW | 589942 | 2% | 94% | 17 |
| 6 | TWO ONE SIDED TESTS PROCEDURE | authKW | 261288 | 1% | 89% | 8 |
| 7 | INTERVAL HYPOTHESIS | authKW | 228937 | 1% | 69% | 9 |
| 8 | SCALED BIOEQUIVALENCE LIMITS | authKW | 220463 | 1% | 100% | 6 |
| 9 | SUBJECT BY FORMULATION INTERACTION | authKW | 183719 | 1% | 100% | 5 |
| 10 | ABSORPTION RATE | authKW | 175076 | 3% | 21% | 23 |
Web of Science journal categories |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | Statistics & Probability | 17357 | 35% | 0% | 293 |
| 2 | Pharmacology & Pharmacy | 8737 | 59% | 0% | 492 |
| 3 | Mathematical & Computational Biology | 5451 | 15% | 0% | 125 |
| 4 | Medical Informatics | 5212 | 10% | 0% | 82 |
| 5 | Medicine, Research & Experimental | 404 | 9% | 0% | 77 |
| 6 | Chemistry, Medicinal | 318 | 6% | 0% | 54 |
| 7 | Public, Environmental & Occupational Health | 300 | 9% | 0% | 75 |
| 8 | Chemistry, Multidisciplinary | 162 | 12% | 0% | 99 |
| 9 | Biology | 61 | 3% | 0% | 26 |
| 10 | Psychology, Mathematical | 47 | 1% | 0% | 5 |
Address terms |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | PHARMACOKINET SERV | 117579 | 0% | 80% | 4 |
| 2 | PHARMALYT INC | 82672 | 0% | 75% | 3 |
| 3 | PHARMACOL CLIN EVALUAT | 78271 | 1% | 30% | 7 |
| 4 | AREA FARM TECNOL | 73488 | 0% | 100% | 2 |
| 5 | PHRMA CMC STAT EXPERT TEAM | 73488 | 0% | 100% | 2 |
| 6 | SPRI STAT | 73488 | 0% | 100% | 2 |
| 7 | REGULATORY AFFAIRS QUAL | 73484 | 0% | 50% | 4 |
| 8 | OFF PHARMACEUT SCI | 68387 | 2% | 10% | 18 |
| 9 | BIOMETR PHARMACOKINET RD | 66137 | 0% | 60% | 3 |
| 10 | BIOEQUIVALENCE | 64687 | 1% | 20% | 9 |
Journals |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | JOURNAL OF BIOPHARMACEUTICAL STATISTICS | 74250 | 5% | 5% | 43 |
| 2 | INTERNATIONAL JOURNAL OF CLINICAL PHARMACOLOGY AND THERAPEUTICS | 50284 | 9% | 2% | 73 |
| 3 | PHARMACEUTICAL STATISTICS | 33832 | 3% | 4% | 22 |
| 4 | STATISTICS IN MEDICINE | 21620 | 8% | 1% | 65 |
| 5 | EUROPEAN JOURNAL OF DRUG METABOLISM AND PHARMACOKINETICS | 13741 | 3% | 2% | 24 |
| 6 | PHARMACEUTICAL RESEARCH | 12387 | 6% | 1% | 52 |
| 7 | AAPS JOURNAL | 11701 | 2% | 2% | 19 |
| 8 | BIOMETRICAL JOURNAL | 8898 | 3% | 1% | 24 |
| 9 | JOURNAL OF PHARMACOKINETICS AND BIOPHARMACEUTICS | 8423 | 2% | 2% | 13 |
| 10 | EUROPEAN JOURNAL OF PHARMACEUTICAL SCIENCES | 4578 | 3% | 1% | 21 |
Author Key Words |
Core articles |
The table includes core articles in the class. The following variables is taken into account for the relevance score of an article in a cluster c: (1) Number of references referring to publications in the class. (2) Share of total number of active references referring to publications in the class. (3) Age of the article. New articles get higher score than old articles. (4) Citation rate, normalized to year. |
| Rank | Reference | # ref. in cl. |
Shr. of ref. in cl. |
Citations |
|---|---|---|---|---|
| 1 | TOTHFALUSI, L , ENDRENYI, L , ARIETA, AG , (2009) EVALUATION OF BIOEQUIVALENCE FOR HIGHLY VARIABLE DRUGS WITH SCALED AVERAGE BIOEQUIVALENCE.CLINICAL PHARMACOKINETICS. VOL. 48. ISSUE 11. P. 725 -743 | 52 | 81% | 21 |
| 2 | KARALIS, V , MACHERAS, P , (2012) CURRENT REGULATORY APPROACHES OF BIOEQUIVALENCE TESTING.EXPERT OPINION ON DRUG METABOLISM & TOXICOLOGY. VOL. 8. ISSUE 8. P. 929 -942 | 43 | 72% | 7 |
| 3 | VAHL, CI , KANG, Q , (2016) EQUIVALENCE CRITERIA FOR THE SAFETY EVALUATION OF A GENETICALLY MODIFIED CROP: A STATISTICAL PERSPECTIVE.JOURNAL OF AGRICULTURAL SCIENCE. VOL. 154. ISSUE 3. P. 383 -406 | 44 | 56% | 0 |
| 4 | KARALIS, V , SYMILLIDES, M , MACHERAS, P , (2011) NOVEL METHODS TO ASSESS BIOEQUIVALENCE.EXPERT OPINION ON DRUG METABOLISM & TOXICOLOGY. VOL. 7. ISSUE 1. P. 79 -88 | 32 | 80% | 4 |
| 5 | ENDRENYI, L , TOTHFALUSI, L , (2012) METRICS FOR THE EVALUATION OF BIOEQUIVALENCE OF MODIFIED-RELEASE FORMULATIONS.AAPS JOURNAL. VOL. 14. ISSUE 4. P. 813 -819 | 26 | 76% | 6 |
| 6 | KARALIS, V , SYMILLIDES, M , MACHERAS, P , (2012) BIOEQUIVALENCE OF HIGHLY VARIABLE DRUGS: A COMPARISON OF THE NEWLY PROPOSED REGULATORY APPROACHES BY FDA AND EMA.PHARMACEUTICAL RESEARCH. VOL. 29. ISSUE 4. P. 1066 -1077 | 20 | 95% | 11 |
| 7 | HARDE, H , AGRAWAL, AK , JAIN, SY , (2015) TETANUS TOXOIDS LOADED GLUCOMANNOSYLATED CHITOSAN BASED NANOHOMING VACCINE ADJUVANT WITH IMPROVED ORAL STABILITY AND IMMUNOSTIMULATORY RESPONSE.PHARMACEUTICAL RESEARCH. VOL. 32. ISSUE 1. P. 122 -143 | 16 | 100% | 4 |
| 8 | DU, LP , CHOI, L , (2015) LIKELIHOOD APPROACH FOR EVALUATING BIOEQUIVALENCE OF HIGHLY VARIABLE DRUGS.PHARMACEUTICAL STATISTICS. VOL. 14. ISSUE 2. P. 82 -94 | 21 | 81% | 0 |
| 9 | FREITAG, G , CZADO, C , MUNK, A , (2007) A NONPARAMETRIC TEST FOR SIMILARITY OF MARGINALS - WITH APPLICATIONS TO THE ASSESSMENT OF POPULATION BIOEQUIVALENCE.JOURNAL OF STATISTICAL PLANNING AND INFERENCE. VOL. 137. ISSUE 3. P. 697 -711 | 26 | 79% | 3 |
| 10 | KARALIS, V , SYMILLIDES, M , MACHERAS, P , (2011) ON THE LEVELING-OFF PROPERTIES OF THE NEW BIOEQUIVALENCE LIMITS FOR HIGHLY VARIABLE DRUGS OF THE EMA GUIDELINE.EUROPEAN JOURNAL OF PHARMACEUTICAL SCIENCES. VOL. 44. ISSUE 4. P. 497-505 | 18 | 100% | 6 |
Classes with closest relation at Level 1 |