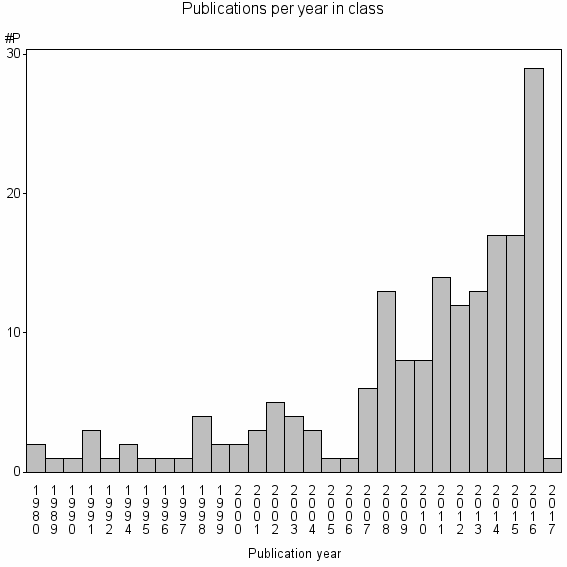
Class information for: |
Basic class information |
| Class id | #P | Avg. number of references |
Database coverage of references |
|---|---|---|---|
| 30020 | 176 | 25.7 | 52% |
Hierarchy of classes |
The table includes all classes above and classes immediately below the current class. |
| Cluster id | Level | Cluster label | #P |
|---|---|---|---|
| 1 | 4 | ECONOMICS//EDUCATION & EDUCATIONAL RESEARCH//PSYCHOL | 3876184 |
| 320 | 3 | AMERICAN JOURNAL OF PHARMACEUTICAL EDUCATION//MEDICATION ERRORS//AMERICAN JOURNAL OF HOSPITAL PHARMACY | 39312 |
| 2978 | 2 | PHARMACOVIGILANCE//ADVERSE DRUG REACTIONS//DRUG SAFETY | 2605 |
| 30020 | 1 | BENEFIT RISK//BENEFIT RISK ASSESSMENT//BENEFIT RISK COMMUNICATION | 176 |
Terms with highest relevance score |
| rank | Term | termType | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|---|
| 1 | BENEFIT RISK | authKW | 1837863 | 14% | 42% | 25 |
| 2 | BENEFIT RISK ASSESSMENT | authKW | 651151 | 10% | 22% | 17 |
| 3 | BENEFIT RISK COMMUNICATION | authKW | 346994 | 1% | 100% | 2 |
| 4 | BENEFIT RISK EVALUAT | address | 346994 | 1% | 100% | 2 |
| 5 | BENEFIT RISK METHODOLOGIES | authKW | 346994 | 1% | 100% | 2 |
| 6 | CUSTOM MADE MEDICINE | authKW | 346994 | 1% | 100% | 2 |
| 7 | GLOBAL REGULATORY AFFAIRS NEUROSCI | address | 346994 | 1% | 100% | 2 |
| 8 | PHARMACOVIGILANCE MED SAFETY | address | 346994 | 1% | 100% | 2 |
| 9 | REGULATORY AFFAIRS EUROPE | address | 346994 | 1% | 100% | 2 |
| 10 | BENEFIT HARM ASSESSMENT | authKW | 231328 | 1% | 67% | 2 |
Web of Science journal categories |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | Medical Informatics | 3337 | 17% | 0% | 30 |
| 2 | Health Care Sciences & Services | 1846 | 22% | 0% | 39 |
| 3 | Statistics & Probability | 1657 | 24% | 0% | 42 |
| 4 | Pharmacology & Pharmacy | 1626 | 56% | 0% | 98 |
| 5 | Mathematical & Computational Biology | 866 | 13% | 0% | 23 |
| 6 | Public, Environmental & Occupational Health | 293 | 18% | 0% | 31 |
| 7 | Health Policy & Services | 236 | 6% | 0% | 11 |
| 8 | Medicine, Research & Experimental | 50 | 7% | 0% | 13 |
| 9 | Toxicology | 20 | 4% | 0% | 7 |
| 10 | Economics | 13 | 4% | 0% | 7 |
Address terms |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | BENEFIT RISK EVALUAT | 346994 | 1% | 100% | 2 |
| 2 | GLOBAL REGULATORY AFFAIRS NEUROSCI | 346994 | 1% | 100% | 2 |
| 3 | PHARMACOVIGILANCE MED SAFETY | 346994 | 1% | 100% | 2 |
| 4 | REGULATORY AFFAIRS EUROPE | 346994 | 1% | 100% | 2 |
| 5 | CLIN DEV BIOSTAT 1 | 231328 | 1% | 67% | 2 |
| 6 | REGULATORY EXCELLENCE | 231328 | 1% | 67% | 2 |
| 7 | AUTON PHARMACOVIGILANCE | 173497 | 1% | 100% | 1 |
| 8 | BENEFIT RISK ASSESSMENT | 173497 | 1% | 100% | 1 |
| 9 | CLIN GENET GENET ECON | 173497 | 1% | 100% | 1 |
| 10 | CLIN REPORTING SYST | 173497 | 1% | 100% | 1 |
Journals |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | THERAPEUTIC INNOVATION & REGULATORY SCIENCE | 154823 | 10% | 5% | 18 |
| 2 | DRUG INFORMATION JOURNAL | 51207 | 10% | 2% | 18 |
| 3 | STATISTICS IN BIOPHARMACEUTICAL RESEARCH | 46059 | 5% | 3% | 9 |
| 4 | PHARMACEUTICAL STATISTICS | 21134 | 5% | 2% | 8 |
| 5 | JOURNAL OF BIOPHARMACEUTICAL STATISTICS | 15358 | 5% | 1% | 9 |
| 6 | PHARMACOEPIDEMIOLOGY AND DRUG SAFETY | 10810 | 7% | 1% | 12 |
| 7 | CLINICAL TRIALS | 3733 | 2% | 1% | 4 |
| 8 | VALUE IN HEALTH | 2781 | 3% | 0% | 5 |
| 9 | DRUG SAFETY | 2072 | 3% | 0% | 5 |
| 10 | CLINICAL PHARMACOLOGY & THERAPEUTICS | 1899 | 5% | 0% | 8 |
Author Key Words |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
LCSH search | Wikipedia search |
|---|---|---|---|---|---|---|---|
| 1 | BENEFIT RISK | 1837863 | 14% | 42% | 25 | Search BENEFIT+RISK | Search BENEFIT+RISK |
| 2 | BENEFIT RISK ASSESSMENT | 651151 | 10% | 22% | 17 | Search BENEFIT+RISK+ASSESSMENT | Search BENEFIT+RISK+ASSESSMENT |
| 3 | BENEFIT RISK COMMUNICATION | 346994 | 1% | 100% | 2 | Search BENEFIT+RISK+COMMUNICATION | Search BENEFIT+RISK+COMMUNICATION |
| 4 | BENEFIT RISK METHODOLOGIES | 346994 | 1% | 100% | 2 | Search BENEFIT+RISK+METHODOLOGIES | Search BENEFIT+RISK+METHODOLOGIES |
| 5 | CUSTOM MADE MEDICINE | 346994 | 1% | 100% | 2 | Search CUSTOM+MADE+MEDICINE | Search CUSTOM+MADE+MEDICINE |
| 6 | BENEFIT HARM ASSESSMENT | 231328 | 1% | 67% | 2 | Search BENEFIT+HARM+ASSESSMENT | Search BENEFIT+HARM+ASSESSMENT |
| 7 | PATHODYNAMICS | 231328 | 1% | 67% | 2 | Search PATHODYNAMICS | Search PATHODYNAMICS |
| 8 | BENEFIT RISK ANALYSIS | 180718 | 3% | 21% | 5 | Search BENEFIT+RISK+ANALYSIS | Search BENEFIT+RISK+ANALYSIS |
| 9 | ADVERSE DRUG REACTION SURVEILLANCE | 173497 | 1% | 100% | 1 | Search ADVERSE+DRUG+REACTION+SURVEILLANCE | Search ADVERSE+DRUG+REACTION+SURVEILLANCE |
| 10 | ADVERSE EVEN IS | 173497 | 1% | 100% | 1 | Search ADVERSE+EVEN+IS | Search ADVERSE+EVEN+IS |
Core articles |
The table includes core articles in the class. The following variables is taken into account for the relevance score of an article in a cluster c: (1) Number of references referring to publications in the class. (2) Share of total number of active references referring to publications in the class. (3) Age of the article. New articles get higher score than old articles. (4) Citation rate, normalized to year. |
| Rank | Reference | # ref. in cl. |
Shr. of ref. in cl. |
Citations |
|---|---|---|---|---|
| 1 | PIGNATTI, F , ASHBY, D , BRASS, EP , EICHLER, HG , FREY, P , HILLEGE, HL , HORI, A , LEVITAN, B , LIBERTI, L , LOEFSTEDT, RE , ET AL (2015) STRUCTURED FRAMEWORKS TO INCREASE THE TRANSPARENCY OF THE ASSESSMENT OF BENEFITS AND RISKS OF MEDICINES: CURRENT STATUS AND POSSIBLE FUTURE DIRECTIONS.CLINICAL PHARMACOLOGY & THERAPEUTICS. VOL. 98. ISSUE 5. P. 522 -533 | 9 | 90% | 5 |
| 2 | WITTES, J , CROWE, B , CHUANG-STEIN, C , GUETTNER, A , HALL, D , JIANG, Q , ODENHEIMER, D , XIA, HA , KRAMER, J , (2015) THE FDA'S FINAL RULE ON EXPEDITED SAFETY REPORTING: STATISTICAL CONSIDERATIONS.STATISTICS IN BIOPHARMACEUTICAL RESEARCH. VOL. 7. ISSUE 3. P. 174 -190 | 9 | 90% | 2 |
| 3 | MT-ISA, S , OUWENS, M , ROBERT, V , GEBEL, M , SCHACHT, A , HIRSCH, I , (2016) STRUCTURED BENEFIT-RISK ASSESSMENT: A REVIEW OF KEY PUBLICATIONS AND INITIATIVES ON FRAMEWORKS AND METHODOLOGIES.PHARMACEUTICAL STATISTICS. VOL. 15. ISSUE 4. P. 324 -332 | 10 | 71% | 1 |
| 4 | MT-ISA, S , HALLGREEN, CE , WANG, N , CALLREUS, T , GENOV, G , HIRSCH, I , HOBBIGER, SF , HOCKLEY, KS , LUCIANI, D , PHILLIPS, LD , ET AL (2014) BALANCING BENEFIT AND RISK OF MEDICINES: A SYSTEMATIC REVIEW AND CLASSIFICATION OF AVAILABLE METHODOLOGIES.PHARMACOEPIDEMIOLOGY AND DRUG SAFETY. VOL. 23. ISSUE 7. P. 667 -678 | 23 | 30% | 16 |
| 5 | WALKER, S , MCAUSLANE, N , LIBERTI, L , LEONG, J , SALEK, S , (2015) A UNIVERSAL FRAMEWORK FOR THE BENEFIT-RISK ASSESSMENT OF MEDICINES: IS THIS THE WAY FORWARD?.THERAPEUTIC INNOVATION & REGULATORY SCIENCE. VOL. 49. ISSUE 1. P. 17 -25 | 7 | 100% | 5 |
| 6 | BENDER, R , BECKMANN, L , LANGE, S , (2016) BIOMETRICAL ISSUES IN THE ANALYSIS OF ADVERSE EVENTS WITHIN THE BENEFIT ASSESSMENT OF DRUGS.PHARMACEUTICAL STATISTICS. VOL. 15. ISSUE 4. P. 292 -296 | 8 | 67% | 1 |
| 7 | CHUANG-STEIN, C , XIA, HA , (2013) THE PRACTICE OF PRE-MARKETING SAFETY ASSESSMENT IN DRUG DEVELOPMENT.JOURNAL OF BIOPHARMACEUTICAL STATISTICS. VOL. 23. ISSUE 1. P. 3 -25 | 9 | 60% | 0 |
| 8 | BUJAR, M , MCAUSLANE, N , SALEK, S , WALKER, S , (2016) QUALITY OF REGULATORY DECISION-MAKING PRACTICES: ISSUES FACING COMPANIES AND AGENCIES.THERAPEUTIC INNOVATION & REGULATORY SCIENCE. VOL. 50. ISSUE 4. P. 487 -495 | 5 | 100% | 0 |
| 9 | MA, HJ , JIANG, Q , CHUANG-STEIN, C , EVANS, SR , HE, WL , QUARTEY, G , SCOTT, J , WEN, SH , ARANI, R , (2016) CONSIDERATIONS ON ENDPOINT SELECTION, WEIGHTING DETERMINATION, AND UNCERTAINTY EVALUATION IN THE BENEFIT-RISK ASSESSMENT OF MEDICAL PRODUCT.STATISTICS IN BIOPHARMACEUTICAL RESEARCH. VOL. 8. ISSUE 4. P. 417 -425 | 11 | 42% | 0 |
| 10 | DAVIS, B , SOUTHWORTH, H , (2016) STATISTICAL ANALYSIS OF CUMULATIVE SERIOUS ADVERSE EVENT DATA FROM DEVELOPMENT SAFETY UPDATE REPORTS.THERAPEUTIC INNOVATION & REGULATORY SCIENCE. VOL. 50. ISSUE 2. P. 188 -194 | 7 | 64% | 0 |
Classes with closest relation at Level 1 |