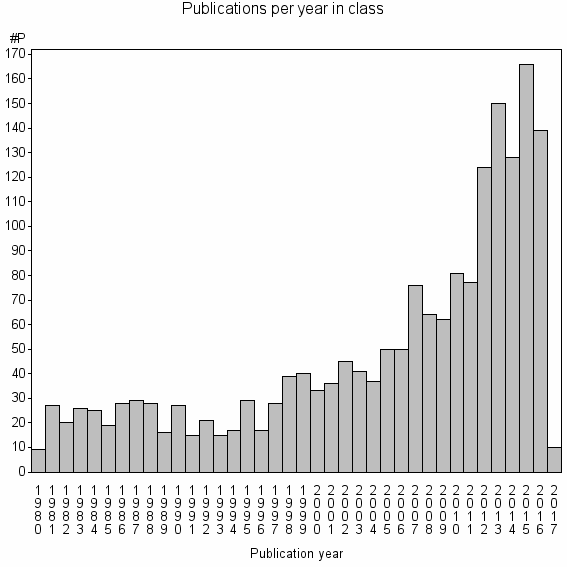
Class information for: |
Basic class information |
| Class id | #P | Avg. number of references |
Database coverage of references |
|---|---|---|---|
| 4114 | 1844 | 25.6 | 57% |
Hierarchy of classes |
The table includes all classes above and classes immediately below the current class. |
| Cluster id | Level | Cluster label | #P |
|---|---|---|---|
| 1 | 4 | ECONOMICS//EDUCATION & EDUCATIONAL RESEARCH//PSYCHOL | 3876184 |
| 320 | 3 | AMERICAN JOURNAL OF PHARMACEUTICAL EDUCATION//MEDICATION ERRORS//AMERICAN JOURNAL OF HOSPITAL PHARMACY | 39312 |
| 2978 | 2 | PHARMACOVIGILANCE//ADVERSE DRUG REACTIONS//DRUG SAFETY | 2605 |
| 4114 | 1 | PHARMACOVIGILANCE//ADVERSE DRUG REACTIONS//DRUG SAFETY | 1844 |
Terms with highest relevance score |
| rank | Term | termType | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|---|
| 1 | PHARMACOVIGILANCE | authKW | 2306129 | 21% | 36% | 385 |
| 2 | ADVERSE DRUG REACTIONS | authKW | 801366 | 19% | 14% | 347 |
| 3 | DRUG SAFETY | journal | 658817 | 16% | 14% | 288 |
| 4 | SPONTANEOUS REPORTING | authKW | 635908 | 4% | 59% | 65 |
| 5 | PHARMACOEPIDEMIOLOGY AND DRUG SAFETY | journal | 229731 | 10% | 8% | 179 |
| 6 | ADVERSE DRUG REACTION REPORTING SYSTEMS | authKW | 228127 | 2% | 33% | 42 |
| 7 | SPONTANEOUS REPORTING SYSTEM | authKW | 220157 | 1% | 53% | 25 |
| 8 | ADR REPORTING | authKW | 216350 | 1% | 93% | 14 |
| 9 | RISK MANAGEMENT STRATEGY | address | 202827 | 1% | 88% | 14 |
| 10 | UPPSALA MONITORING | address | 186337 | 1% | 49% | 23 |
Web of Science journal categories |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | Pharmacology & Pharmacy | 24779 | 67% | 0% | 1227 |
| 2 | Public, Environmental & Occupational Health | 9993 | 31% | 0% | 563 |
| 3 | Toxicology | 6349 | 18% | 0% | 332 |
| 4 | Medical Informatics | 5830 | 7% | 0% | 130 |
| 5 | Health Care Sciences & Services | 2000 | 7% | 0% | 137 |
| 6 | Medicine, General & Internal | 776 | 11% | 0% | 202 |
| 7 | Mathematical & Computational Biology | 393 | 3% | 0% | 55 |
| 8 | Computer Science, Interdisciplinary Applications | 172 | 3% | 0% | 60 |
| 9 | Statistics & Probability | 154 | 3% | 0% | 51 |
| 10 | Computer Science, Information Systems | 80 | 2% | 0% | 43 |
Address terms |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | RISK MANAGEMENT STRATEGY | 202827 | 1% | 88% | 14 |
| 2 | UPPSALA MONITORING | 186337 | 1% | 49% | 23 |
| 3 | PHARM PHARMACOTHER Y PHARMACEUT CARE | 125210 | 1% | 69% | 11 |
| 4 | ABORATING INT DRUG MONITORING | 108667 | 1% | 41% | 16 |
| 5 | LESIM | 81508 | 0% | 62% | 8 |
| 6 | INTEGRAT EDUC PHARM PHARMACEUT SCI | 75685 | 0% | 57% | 8 |
| 7 | OBSERVAT MED OUTCOMES PARTNERSHIP | 59131 | 0% | 71% | 5 |
| 8 | WHO ABORATING INT DRUG MONITORING | 57944 | 0% | 50% | 7 |
| 9 | LAREB | 52983 | 0% | 80% | 4 |
| 10 | NO POLYTECH HLTH | 52983 | 0% | 80% | 4 |
Journals |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | DRUG SAFETY | 658817 | 16% | 14% | 288 |
| 2 | PHARMACOEPIDEMIOLOGY AND DRUG SAFETY | 229731 | 10% | 8% | 179 |
| 3 | THERAPIE | 42697 | 4% | 3% | 81 |
| 4 | DRUG INFORMATION JOURNAL | 27825 | 2% | 4% | 43 |
| 5 | ADVERSE DRUG REACTIONS AND TOXICOLOGICAL REVIEWS | 16965 | 1% | 7% | 14 |
| 6 | BRITISH JOURNAL OF CLINICAL PHARMACOLOGY | 12016 | 4% | 1% | 77 |
| 7 | EUROPEAN JOURNAL OF CLINICAL PHARMACOLOGY | 10433 | 4% | 1% | 67 |
| 8 | INDIAN JOURNAL OF PHARMACOLOGY | 7461 | 1% | 2% | 22 |
| 9 | AMERICAN JOURNAL OF HOSPITAL PHARMACY | 6955 | 2% | 1% | 32 |
| 10 | EXPERT OPINION ON DRUG SAFETY | 5738 | 1% | 2% | 19 |
Author Key Words |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
LCSH search | Wikipedia search |
|---|---|---|---|---|---|---|---|
| 1 | PHARMACOVIGILANCE | 2306129 | 21% | 36% | 385 | Search PHARMACOVIGILANCE | Search PHARMACOVIGILANCE |
| 2 | ADVERSE DRUG REACTIONS | 801366 | 19% | 14% | 347 | Search ADVERSE+DRUG+REACTIONS | Search ADVERSE+DRUG+REACTIONS |
| 3 | SPONTANEOUS REPORTING | 635908 | 4% | 59% | 65 | Search SPONTANEOUS+REPORTING | Search SPONTANEOUS+REPORTING |
| 4 | ADVERSE DRUG REACTION REPORTING SYSTEMS | 228127 | 2% | 33% | 42 | Search ADVERSE+DRUG+REACTION+REPORTING+SYSTEMS | Search ADVERSE+DRUG+REACTION+REPORTING+SYSTEMS |
| 5 | SPONTANEOUS REPORTING SYSTEM | 220157 | 1% | 53% | 25 | Search SPONTANEOUS+REPORTING+SYSTEM | Search SPONTANEOUS+REPORTING+SYSTEM |
| 6 | ADR REPORTING | 216350 | 1% | 93% | 14 | Search ADR+REPORTING | Search ADR+REPORTING |
| 7 | PATIENT REPORTING | 170303 | 1% | 86% | 12 | Search PATIENT+REPORTING | Search PATIENT+REPORTING |
| 8 | MEDDRA | 169538 | 1% | 64% | 16 | Search MEDDRA | Search MEDDRA |
| 9 | DISPROPORTIONALITY ANALYSIS | 162256 | 1% | 70% | 14 | Search DISPROPORTIONALITY+ANALYSIS | Search DISPROPORTIONALITY+ANALYSIS |
| 10 | PROPORTIONAL REPORTING RATIO | 149018 | 0% | 100% | 9 | Search PROPORTIONAL+REPORTING+RATIO | Search PROPORTIONAL+REPORTING+RATIO |
Core articles |
The table includes core articles in the class. The following variables is taken into account for the relevance score of an article in a cluster c: (1) Number of references referring to publications in the class. (2) Share of total number of active references referring to publications in the class. (3) Age of the article. New articles get higher score than old articles. (4) Citation rate, normalized to year. |
| Rank | Reference | # ref. in cl. |
Shr. of ref. in cl. |
Citations |
|---|---|---|---|---|
| 1 | BATE, A , EVANS, SJW , (2009) QUANTITATIVE SIGNAL DETECTION USING SPONTANEOUS ADR REPORTING.PHARMACOEPIDEMIOLOGY AND DRUG SAFETY. VOL. 18. ISSUE 6. P. 427-436 | 49 | 89% | 160 |
| 2 | SAKAEDA, T , TAMON, A , KADOYAMA, K , OKUNO, Y , (2013) DATA MINING OF THE PUBLIC VERSION OF THE FDA ADVERSE EVENT REPORTING SYSTEM.INTERNATIONAL JOURNAL OF MEDICAL SCIENCES. VOL. 10. ISSUE 7. P. 796-803 | 54 | 72% | 49 |
| 3 | WISNIEWSKI, AFZ , BATE, A , BOUSQUET, C , BRUECKNER, A , CANDORE, G , JUHLIN, K , MACIA-MARTINEZ, MA , MANLIK, K , QUARCOO, N , SEABROKE, S , ET AL (2016) GOOD SIGNAL DETECTION PRACTICES: EVIDENCE FROM IMI PROTECT.DRUG SAFETY. VOL. 39. ISSUE 6. P. 469 -490 | 48 | 92% | 0 |
| 4 | LOPEZ-GONZALEZ, E , HERDEIRO, MT , FIGUEIRAS, A , (2009) DETERMINANTS OF UNDER-REPORTING OF ADVERSE DRUG REACTIONS A SYSTEMATIC REVIEW.DRUG SAFETY. VOL. 32. ISSUE 1. P. 19-31 | 39 | 93% | 127 |
| 5 | KOUTKIAS, VG , JAULENT, MC , (2015) COMPUTATIONAL APPROACHES FOR PHARMACOVIGILANCE SIGNAL DETECTION: TOWARD INTEGRATED AND SEMANTICALLY-ENRICHED FRAMEWORKS.DRUG SAFETY. VOL. 38. ISSUE 3. P. 219 -232 | 42 | 89% | 5 |
| 6 | INACIO, P , CAVACO, A , AIRAKSINEN, M , (2017) THE VALUE OF PATIENT REPORTING TO THE PHARMACOVIGILANCE SYSTEM: A SYSTEMATIC REVIEW.BRITISH JOURNAL OF CLINICAL PHARMACOLOGY. VOL. 83. ISSUE 2. P. 227 -246 | 42 | 91% | 0 |
| 7 | GONZALEZ-GONZALEZ, C , LOPEZ-GONZALEZ, E , HERDEIRO, MT , FIGUEIRAS, A , (2013) STRATEGIES TO IMPROVE ADVERSE DRUG REACTION REPORTING: A CRITICAL AND SYSTEMATIC REVIEW.DRUG SAFETY. VOL. 36. ISSUE 5. P. 317 -328 | 42 | 84% | 17 |
| 8 | VARALLO, FR , GUIMARAES, SDP , ABJAUDE, SAR , MASTROIANNI, PD , (2014) CAUSES FOR THE UNDERREPORTING OF ADVERSE DRUG EVENTS BY HEALTH PROFESSIONALS: A SYSTEMATIC REVIEW.REVISTA DA ESCOLA DE ENFERMAGEM DA USP. VOL. 48. ISSUE 4. P. 739 -747 | 39 | 91% | 7 |
| 9 | HARMARK, L , VAN HUNSEL, F , GRUNDMARK, B , (2015) ADR REPORTING BY THE GENERAL PUBLIC: LESSONS LEARNT FROM THE DUTCH AND SWEDISH SYSTEMS.DRUG SAFETY. VOL. 38. ISSUE 4. P. 337 -347 | 28 | 93% | 6 |
| 10 | HAZELL, L , SHAKIR, SAW , (2006) UNDER-REPORTING OF ADVERSE DRUG REACTIONS - A SYSTEMATIC REVIEW.DRUG SAFETY. VOL. 29. ISSUE 5. P. 385-396 | 30 | 73% | 398 |
Classes with closest relation at Level 1 |