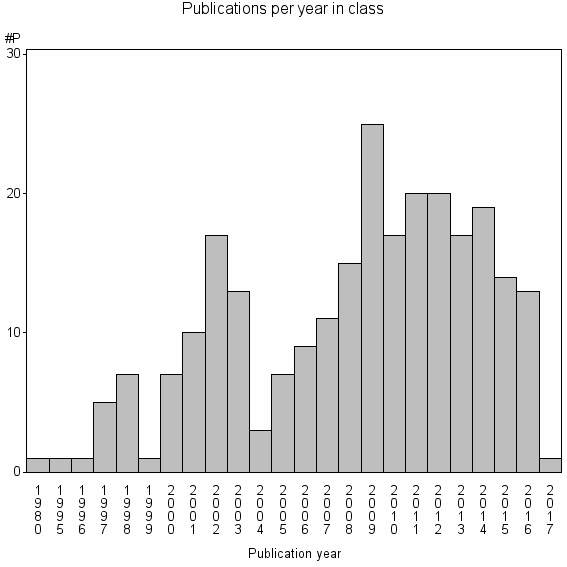
Class information for: |
Basic class information |
| Class id | #P | Avg. number of references |
Database coverage of references |
|---|---|---|---|
| 26445 | 254 | 20.7 | 43% |
Hierarchy of classes |
The table includes all classes above and classes immediately below the current class. |
| Cluster id | Level | Cluster label | #P |
|---|---|---|---|
| 1 | 4 | ECONOMICS//EDUCATION & EDUCATIONAL RESEARCH//PSYCHOL | 3876184 |
| 320 | 3 | AMERICAN JOURNAL OF PHARMACEUTICAL EDUCATION//MEDICATION ERRORS//AMERICAN JOURNAL OF HOSPITAL PHARMACY | 39312 |
| 1464 | 2 | FOOD AND DRUG LAW JOURNAL//CONFLICT OF INTEREST//ORPHAN DRUGS | 7747 |
| 26445 | 1 | DRUG LAG//JAPANESE CLINICAL TRIALS//GLOBAL DRUG DEVELOPMENT | 254 |
Terms with highest relevance score |
| rank | Term | termType | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|---|
| 1 | DRUG LAG | authKW | 808122 | 4% | 61% | 11 |
| 2 | JAPANESE CLINICAL TRIALS | authKW | 480871 | 2% | 100% | 4 |
| 3 | GLOBAL DRUG DEVELOPMENT | authKW | 429346 | 2% | 71% | 5 |
| 4 | OFF PHARMACEUT IND | address | 429346 | 2% | 71% | 5 |
| 5 | CLIN TRIAL GUIDANCE | address | 360653 | 1% | 100% | 3 |
| 6 | REVIEW AUDIT | address | 360653 | 1% | 100% | 3 |
| 7 | PHARMACEUT REGULATORY SCI | address | 334509 | 3% | 35% | 8 |
| 8 | ETHNIC FACTORS | authKW | 274772 | 3% | 29% | 8 |
| 9 | STUDY DRUG DEV | address | 243427 | 4% | 23% | 9 |
| 10 | DRUG INFORMATION JOURNAL | journal | 241989 | 19% | 4% | 47 |
Web of Science journal categories |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | Pharmacology & Pharmacy | 3413 | 67% | 0% | 169 |
| 2 | Health Care Sciences & Services | 2833 | 23% | 0% | 58 |
| 3 | Medical Informatics | 416 | 5% | 0% | 13 |
| 4 | Health Policy & Services | 102 | 4% | 0% | 9 |
| 5 | Medicine, Research & Experimental | 65 | 7% | 0% | 18 |
| 6 | Oncology | 52 | 9% | 0% | 24 |
| 7 | Biotechnology & Applied Microbiology | 28 | 6% | 0% | 15 |
| 8 | Integrative & Complementary Medicine | 19 | 1% | 0% | 3 |
| 9 | Ethics | 11 | 1% | 0% | 2 |
| 10 | Medicine, General & Internal | 10 | 5% | 0% | 12 |
Address terms |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | OFF PHARMACEUT IND | 429346 | 2% | 71% | 5 |
| 2 | CLIN TRIAL GUIDANCE | 360653 | 1% | 100% | 3 |
| 3 | REVIEW AUDIT | 360653 | 1% | 100% | 3 |
| 4 | PHARMACEUT REGULATORY SCI | 334509 | 3% | 35% | 8 |
| 5 | STUDY DRUG DEV | 243427 | 4% | 23% | 9 |
| 6 | ONCOLCHUO KU | 240436 | 1% | 100% | 2 |
| 7 | PHARMACEUT MED DEVICES EVALUAT | 154556 | 2% | 21% | 6 |
| 8 | ABT CHEM BIOL CHEM | 120218 | 0% | 100% | 1 |
| 9 | BEIJING CHINESE MAT MEDICA QUAL EVALUAT | 120218 | 0% | 100% | 1 |
| 10 | BUNKYO GREEN COURT OFF | 120218 | 0% | 100% | 1 |
Journals |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | DRUG INFORMATION JOURNAL | 241989 | 19% | 4% | 47 |
| 2 | THERAPEUTIC INNOVATION & REGULATORY SCIENCE | 55944 | 5% | 4% | 13 |
| 3 | CLINICAL RESEARCH AND REGULATORY AFFAIRS | 28344 | 2% | 5% | 5 |
| 4 | CONTEMPORARY CLINICAL TRIALS | 5476 | 3% | 1% | 8 |
| 5 | CLINICAL PHARMACOLOGY & THERAPEUTICS | 5274 | 6% | 0% | 16 |
| 6 | JOURNAL OF CLINICAL PHARMACY AND THERAPEUTICS | 4530 | 4% | 0% | 9 |
| 7 | NAGOYA JOURNAL OF MEDICAL SCIENCE | 3484 | 1% | 1% | 3 |
| 8 | JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES | 2776 | 2% | 1% | 4 |
| 9 | YAKUGAKU ZASSHI-JOURNAL OF THE PHARMACEUTICAL SOCIETY OF JAPAN | 1859 | 4% | 0% | 9 |
| 10 | JOURNAL OF BIOPHARMACEUTICAL STATISTICS | 1178 | 1% | 0% | 3 |
Author Key Words |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
LCSH search | Wikipedia search |
|---|---|---|---|---|---|---|---|
| 1 | DRUG LAG | 808122 | 4% | 61% | 11 | Search DRUG+LAG | Search DRUG+LAG |
| 2 | JAPANESE CLINICAL TRIALS | 480871 | 2% | 100% | 4 | Search JAPANESE+CLINICAL+TRIALS | Search JAPANESE+CLINICAL+TRIALS |
| 3 | GLOBAL DRUG DEVELOPMENT | 429346 | 2% | 71% | 5 | Search GLOBAL+DRUG+DEVELOPMENT | Search GLOBAL+DRUG+DEVELOPMENT |
| 4 | ETHNIC FACTORS | 274772 | 3% | 29% | 8 | Search ETHNIC+FACTORS | Search ETHNIC+FACTORS |
| 5 | ASIAN CLINICAL TRIAL NETWORK | 240436 | 1% | 100% | 2 | Search ASIAN+CLINICAL+TRIAL+NETWORK | Search ASIAN+CLINICAL+TRIAL+NETWORK |
| 6 | CLINICAL GRANT | 240436 | 1% | 100% | 2 | Search CLINICAL+GRANT | Search CLINICAL+GRANT |
| 7 | DRUG MASTER FILE | 240436 | 1% | 100% | 2 | Search DRUG+MASTER+FILE | Search DRUG+MASTER+FILE |
| 8 | FDA MODERNIZATION ACT OF 1997 | 240436 | 1% | 100% | 2 | Search FDA+MODERNIZATION+ACT+OF+1997 | Search FDA+MODERNIZATION+ACT+OF+1997 |
| 9 | FOOD AND DRUG ADMINISTRATION MODERNIZATION | 240436 | 1% | 100% | 2 | Search FOOD+AND+DRUG+ADMINISTRATION+MODERNIZATION | Search FOOD+AND+DRUG+ADMINISTRATION+MODERNIZATION |
| 10 | GDUFA | 240436 | 1% | 100% | 2 | Search GDUFA | Search GDUFA |
Core articles |
The table includes core articles in the class. The following variables is taken into account for the relevance score of an article in a cluster c: (1) Number of references referring to publications in the class. (2) Share of total number of active references referring to publications in the class. (3) Age of the article. New articles get higher score than old articles. (4) Citation rate, normalized to year. |
| Rank | Reference | # ref. in cl. |
Shr. of ref. in cl. |
Citations |
|---|---|---|---|---|
| 1 | MAEDA, H , KUROKAWA, T , (2015) REGULATORY REVIEW TIME FOR APPROVAL OF ONCOLOGY DRUGS IN JAPAN BETWEEN 2001 AND 2014. CONSIDERATIONS OF CHANGES, FACTORS THAT AFFECT REVIEW TIME, AND DIFFERENCE WITH THE UNITED STATES.JOURNAL OF CLINICAL PHARMACOLOGY. VOL. 55. ISSUE 5. P. 481 -489 | 10 | 91% | 2 |
| 2 | MAEDA, H , KUROKAWA, T , (2015) RECENT TRENDS FOR DRUG LAG IN CLINICAL DEVELOPMENT OF ONCOLOGY DRUGS IN JAPAN: DOES THE ONCOLOGY DRUG LAG STILL EXIST IN JAPAN?.INTERNATIONAL JOURNAL OF CLINICAL ONCOLOGY. VOL. 20. ISSUE 6. P. 1072 -1080 | 12 | 67% | 1 |
| 3 | NAKAJIMA, K , DAGHER, R , STRAWN, L , URUSHIDANI, J , KUROKAWA, T , CHIBA, K , (2015) THE RELATIONSHIP BETWEEN DEVELOPMENT START LAG AND APPROVAL LAG IN ONCOLOGY DRUG DEVELOPMENT IN JAPAN.THERAPEUTIC INNOVATION & REGULATORY SCIENCE. VOL. 49. ISSUE 6. P. 911 -919 | 8 | 89% | 0 |
| 4 | ARNOLD, FL , FUKUNAGA, S , KUSAMA, M , MATSUKI, N , ONO, S , (2014) ASSESSMENT OF FACTORS ASSOCIATED WITH DOSE DIFFERENCES BETWEEN JAPAN AND THE UNITED STATES.CLINICAL PHARMACOLOGY & THERAPEUTICS. VOL. 95. ISSUE 5. P. 542-549 | 12 | 57% | 4 |
| 5 | NAKAJIMA, K , CHIBA, K , TSUBAMOTO, H , WALSH, J , STRAWN, L , SUWA, T , (2013) POTENTIAL FACTORS CORRELATING TO THE PMDA'S DECISION TO WAIVE JAPANESE PHASE 2 AND 3 STUDIES FOR ONCOLOGY DRUGS NEW DRUG APPLICATION IN JAPAN.INVESTIGATIONAL NEW DRUGS. VOL. 31. ISSUE 4. P. 1051-1055 | 7 | 100% | 0 |
| 6 | OHWAKI, K , NAKABAYASHI, T , (2014) RELATIONSHIP BETWEEN DRUG LAG AND FACTORS ASSOCIATED WITH CLINICAL TRIALS IN JAPAN.JOURNAL OF CLINICAL PHARMACY AND THERAPEUTICS. VOL. 39. ISSUE 6. P. 649 -652 | 6 | 100% | 2 |
| 7 | ISHIBASHI, T , KUSAMA, M , SUGIYAMA, Y , ONO, S , (2012) ANALYSIS OF REGULATORY REVIEW TIMES OF NEW DRUGS IN JAPAN: ASSOCIATION WITH CHARACTERISTICS OF NEW DRUG APPLICATIONS, REGULATORY AGENCY, AND PHARMACEUTICAL COMPANIES.JOURNAL OF CLINICAL PHARMACY AND THERAPEUTICS. VOL. 37. ISSUE 6. P. 657-663 | 9 | 69% | 3 |
| 8 | SHIMAZAWA, R , IKEDA, M , (2011) DELAYS IN NEUROLOGICAL DRUG DEVELOPMENT IN JAPAN.INTERNAL MEDICINE. VOL. 50. ISSUE 15. P. 1565-1568 | 6 | 100% | 3 |
| 9 | TANIMOTO, T , (2015) A PERSPECTIVE ON THE BENEFIT-RISK ASSESSMENT FOR NEW AND EMERGING PHARMACEUTICALS IN JAPAN.DRUG DESIGN DEVELOPMENT AND THERAPY. VOL. 9. ISSUE . P. 1877 -1888 | 12 | 43% | 0 |
| 10 | SHIMAZAWA, R , KUSUMI, I , IKEDA, M , (2012) DELAYS IN PSYCHIATRIC DRUG DEVELOPMENT IN JAPAN.JOURNAL OF CLINICAL PHARMACY AND THERAPEUTICS. VOL. 37. ISSUE 3. P. 348-351 | 9 | 60% | 4 |
Classes with closest relation at Level 1 |