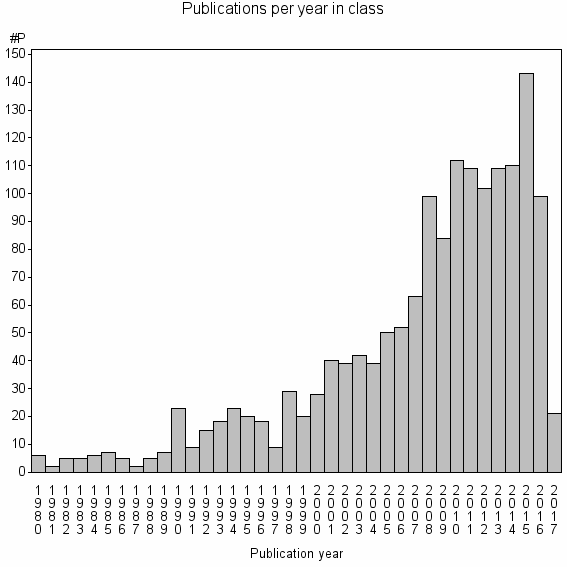
Class information for: |
Basic class information |
| Class id | #P | Avg. number of references |
Database coverage of references |
|---|---|---|---|
| 3420 | 1575 | 26.3 | 64% |
Hierarchy of classes |
The table includes all classes above and classes immediately below the current class. |
| Cluster id | Level | Cluster label | #P |
|---|---|---|---|
| 1 | 4 | ECONOMICS//EDUCATION & EDUCATIONAL RESEARCH//PSYCHOL | 3876184 |
| 499 | 3 | MEDICAL ETHICS//INFORMED CONSENT//ETHICS | 22534 |
| 3420 | 2 | CONTINUAL REASSESSMENT METHOD//DOSE FINDING//PHASE I TRIAL | 1575 |
| 13262 | 1 | CONTINUAL REASSESSMENT METHOD//DOSE FINDING//PHASE I TRIAL | 850 |
| 28722 | 1 | MANAGEMENT HEALTHCARE//ACADEMIC ASSOCIATE PROGRAM//CLINICAL RESEARCH MANAGEMENT | 201 |
| 29589 | 1 | HRDG//ACADEMIC CLINICAL COLLABORATION//ACADEMIC CLINICAL RELATIONSHIPS | 183 |
| 31688 | 1 | CLINICAL COOPERATIVE GROUPS//COMMUNITY CLINICAL ONCOLOGY PROGRAM//CCOP | 149 |
| 33716 | 1 | TARGET BASED DRUG//FULL TIME RESEARCHERS//ACADEMY OF MEDICINE | 121 |
| 36921 | 1 | WICHITA CLIN TRIALS UNIT//AIRWAY SPLINT//BIOMACROMOLECULE PROTEIN | 71 |
Terms with highest relevance score |
| rank | Term | termType | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|---|
| 1 | CONTINUAL REASSESSMENT METHOD | authKW | 1539441 | 6% | 88% | 90 |
| 2 | DOSE FINDING | authKW | 1159702 | 8% | 48% | 124 |
| 3 | PHASE I TRIAL | authKW | 515712 | 7% | 24% | 111 |
| 4 | DOSE FINDING STUDIES | authKW | 447351 | 2% | 77% | 30 |
| 5 | MAXIMUM TOLERATED DOSE | authKW | 378583 | 4% | 30% | 66 |
| 6 | BAYESIAN ADAPTIVE DESIGN | authKW | 216573 | 1% | 62% | 18 |
| 7 | UP AND DOWN DESIGN | authKW | 192712 | 1% | 76% | 13 |
| 8 | ESCALATION WITH OVERDOSE CONTROL | authKW | 174472 | 1% | 100% | 9 |
| 9 | ADAPTIVE DESIGN | authKW | 158617 | 4% | 13% | 65 |
| 10 | TRANSLAT PL STAT | address | 151973 | 1% | 56% | 14 |
Web of Science journal categories |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | Statistics & Probability | 11711 | 21% | 0% | 335 |
| 2 | Medical Informatics | 8700 | 9% | 0% | 146 |
| 3 | Mathematical & Computational Biology | 7453 | 13% | 0% | 202 |
| 4 | Oncology | 4223 | 28% | 0% | 439 |
| 5 | Health Care Sciences & Services | 3392 | 10% | 0% | 162 |
| 6 | Medicine, Research & Experimental | 3078 | 17% | 0% | 272 |
| 7 | Education, Scientific Disciplines | 1997 | 6% | 0% | 89 |
| 8 | Health Policy & Services | 822 | 4% | 0% | 63 |
| 9 | Public, Environmental & Occupational Health | 810 | 10% | 0% | 165 |
| 10 | Pharmacology & Pharmacy | 587 | 14% | 0% | 214 |
Address terms |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | TRANSLAT PL STAT | 151973 | 1% | 56% | 14 |
| 2 | MANAGEMENT HEALTHCARE | 63439 | 0% | 55% | 6 |
| 3 | UNIT MOL THER Y CANC | 58157 | 0% | 100% | 3 |
| 4 | UNIT EA2694 | 43617 | 0% | 75% | 3 |
| 5 | CNRS UMR8203 | 38772 | 0% | 100% | 2 |
| 6 | DATA HLTH CARE GEN INTERNAL MED | 38772 | 0% | 100% | 2 |
| 7 | DEV REGULATORY SERV | 38772 | 0% | 100% | 2 |
| 8 | EQUIPE 22UNIV PARIS 5U1138 | 38772 | 0% | 100% | 2 |
| 9 | HRDG | 38772 | 0% | 100% | 2 |
| 10 | INVESTIGATOR SUPPORT INTEGRAT SERV | 38772 | 0% | 100% | 2 |
Journals |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | STATISTICS IN MEDICINE | 40847 | 8% | 2% | 123 |
| 2 | CLINICAL TRIALS | 35697 | 2% | 5% | 37 |
| 3 | CTS-CLINICAL AND TRANSLATIONAL SCIENCE | 27681 | 2% | 5% | 28 |
| 4 | PHARMACEUTICAL STATISTICS | 23031 | 2% | 5% | 25 |
| 5 | JOURNAL OF BIOPHARMACEUTICAL STATISTICS | 20323 | 2% | 3% | 31 |
| 6 | ACADEMIC MEDICINE | 15720 | 4% | 1% | 70 |
| 7 | CONTEMPORARY CLINICAL TRIALS | 10794 | 2% | 2% | 28 |
| 8 | BIOMETRICS | 10256 | 3% | 1% | 49 |
| 9 | CLINICAL CANCER RESEARCH | 4471 | 4% | 0% | 59 |
| 10 | STATISTICS IN BIOPHARMACEUTICAL RESEARCH | 3101 | 0% | 2% | 7 |
Author Key Words |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
LCSH search | Wikipedia search |
|---|---|---|---|---|---|---|---|
| 1 | CONTINUAL REASSESSMENT METHOD | 1539441 | 6% | 88% | 90 | Search CONTINUAL+REASSESSMENT+METHOD | Search CONTINUAL+REASSESSMENT+METHOD |
| 2 | DOSE FINDING | 1159702 | 8% | 48% | 124 | Search DOSE+FINDING | Search DOSE+FINDING |
| 3 | PHASE I TRIAL | 515712 | 7% | 24% | 111 | Search PHASE+I+TRIAL | Search PHASE+I+TRIAL |
| 4 | DOSE FINDING STUDIES | 447351 | 2% | 77% | 30 | Search DOSE+FINDING+STUDIES | Search DOSE+FINDING+STUDIES |
| 5 | MAXIMUM TOLERATED DOSE | 378583 | 4% | 30% | 66 | Search MAXIMUM+TOLERATED+DOSE | Search MAXIMUM+TOLERATED+DOSE |
| 6 | BAYESIAN ADAPTIVE DESIGN | 216573 | 1% | 62% | 18 | Search BAYESIAN+ADAPTIVE+DESIGN | Search BAYESIAN+ADAPTIVE+DESIGN |
| 7 | UP AND DOWN DESIGN | 192712 | 1% | 76% | 13 | Search UP+AND+DOWN+DESIGN | Search UP+AND+DOWN+DESIGN |
| 8 | ESCALATION WITH OVERDOSE CONTROL | 174472 | 1% | 100% | 9 | Search ESCALATION+WITH+OVERDOSE+CONTROL | Search ESCALATION+WITH+OVERDOSE+CONTROL |
| 9 | ADAPTIVE DESIGN | 158617 | 4% | 13% | 65 | Search ADAPTIVE+DESIGN | Search ADAPTIVE+DESIGN |
| 10 | RESEARCH BIOPSY | 142747 | 1% | 82% | 9 | Search RESEARCH+BIOPSY | Search RESEARCH+BIOPSY |
Core articles |
The table includes core articles in the class. The following variables is taken into account for the relevance score of an article in a cluster c: (1) Number of references referring to publications in the class. (2) Share of total number of active references referring to publications in the class. (3) Age of the article. New articles get higher score than old articles. (4) Citation rate, normalized to year. |
| Rank | Reference | # ref. in cl. |
Shr. of ref. in cl. |
Citations |
|---|---|---|---|---|
| 1 | HARRINGTON, JA , WHEELER, GM , SWEETING, MJ , MANDER, AP , JODRELL, DI , (2013) ADAPTIVE DESIGNS FOR DUAL-AGENT PHASE I DOSE-ESCALATION STUDIES.NATURE REVIEWS CLINICAL ONCOLOGY. VOL. 10. ISSUE 5. P. 277 -288 | 61 | 85% | 16 |
| 2 | DOUSSAU, A , GEOERGER, B , JIMENEZ, I , PAOLETTI, X , (2016) INNOVATIONS FOR PHASE I DOSE-FINDING DESIGNS IN PEDIATRIC ONCOLOGY CLINICAL TRIALS.CONTEMPORARY CLINICAL TRIALS. VOL. 47. ISSUE . P. 217 -227 | 40 | 83% | 2 |
| 3 | POTTER, DM , (2006) PHASE I STUDIES OF CHEMOTHERAPEUTIC AGENTS IN CANCER PATIENTS: A REVIEW OF THE DESIGNS.JOURNAL OF BIOPHARMACEUTICAL STATISTICS. VOL. 16. ISSUE 5. P. 579-604 | 55 | 85% | 17 |
| 4 | PETRONI, GR , WAGES, NA , PAUX, G , DUBOIS, F , (2017) IMPLEMENTATION OF ADAPTIVE METHODS IN EARLY-PHASE CLINICAL TRIALS.STATISTICS IN MEDICINE. VOL. 36. ISSUE 2. P. 215 -224 | 25 | 89% | 1 |
| 5 | WAGES, NA , IVANOVA, A , MARCHENKO, O , (2016) PRACTICAL DESIGNS FOR PHASE I COMBINATION STUDIES IN ONCOLOGY.JOURNAL OF BIOPHARMACEUTICAL STATISTICS. VOL. 26. ISSUE 1. P. 150 -166 | 35 | 92% | 0 |
| 6 | HORTON, BJ , WAGES, NA , CONAWAY, MR , (2017) PERFORMANCE OF TOXICITY PROBABILITY INTERVAL BASED DESIGNS IN CONTRAST TO THE CONTINUAL REASSESSMENT METHOD.STATISTICS IN MEDICINE. VOL. 36. ISSUE 2. P. 291 -300 | 19 | 100% | 1 |
| 7 | ORON, AP , HOFF, PD , (2013) SMALL-SAMPLE BEHAVIOR OF NOVEL PHASE I CANCER TRIAL DESIGNS.CLINICAL TRIALS. VOL. 10. ISSUE 1. P. 63 -80 | 36 | 88% | 6 |
| 8 | ZHOU, YH , (2010) ADAPTIVE DESIGNS FOR PHASE I DOSE-FINDING STUDIES.FUNDAMENTAL & CLINICAL PHARMACOLOGY. VOL. 24. ISSUE 2. P. 129 -138 | 36 | 97% | 2 |
| 9 | O'QUIGLEY, J , CONAWAY, M , (2010) CONTINUAL REASSESSMENT AND RELATED DOSE-FINDING DESIGNS.STATISTICAL SCIENCE. VOL. 25. ISSUE 2. P. 202 -216 | 36 | 92% | 9 |
| 10 | NEUENSCHWANDER, B , BRANSON, M , GSPONER, T , (2008) CRITICAL ASPECTS OF THE BAYESIAN APPROACH TO PHASE I CANCER TRIALS.STATISTICS IN MEDICINE. VOL. 27. ISSUE 13. P. 2420-2439 | 31 | 100% | 63 |
Classes with closest relation at Level 2 |