Class information for: |
Basic class information |
| Class id | #P | Avg. number of references |
Database coverage of references |
|---|---|---|---|
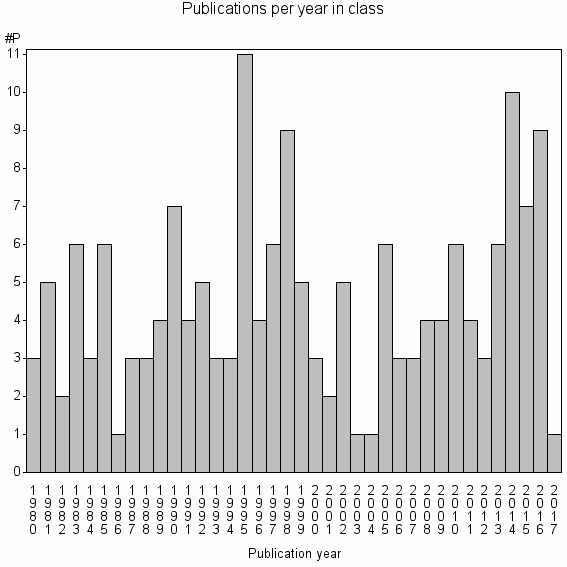
| 30267 | 171 | 27.4 | 51% |
Hierarchy of classes |
The table includes all classes above and classes immediately below the current class. |
| Cluster id | Level | Cluster label | #P |
|---|---|---|---|
| 0 | 4 | BIOCHEMISTRY & MOLECULAR BIOLOGY//CELL BIOLOGY//ONCOLOGY | 4064930 |
| 373 | 3 | ENDOGENOUS RETROVIRUS//VIROLOGY//JOURNAL OF VIROLOGY | 32972 |
| 3655 | 2 | RESIDUAL DNA//AFFINITY RESINS//NONSPECIFIC BINDING PROTEINS | 1185 |
| 30267 | 1 | RESIDUAL DNA//RESIDUAL HOST CELL DNA//DNA ONCOGENICITY | 171 |
Terms with highest relevance score |
| rank | Term | termType | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|---|
| 1 | RESIDUAL DNA | authKW | 1269830 | 5% | 89% | 8 |
| 2 | RESIDUAL HOST CELL DNA | authKW | 401781 | 2% | 75% | 3 |
| 3 | DNA ONCOGENICITY | authKW | 357140 | 1% | 100% | 2 |
| 4 | ECT ANIM PATHOGEN MICROORGANISMS | address | 357140 | 1% | 100% | 2 |
| 5 | HISTORY OF VACCINES | authKW | 357140 | 1% | 100% | 2 |
| 6 | HOST RESIDUAL DNA | authKW | 357140 | 1% | 100% | 2 |
| 7 | RESIDUAL HOST DNA | authKW | 357140 | 1% | 100% | 2 |
| 8 | MERCK THER EUT | address | 229587 | 2% | 43% | 3 |
| 9 | 3D VIRUS CULTURE | authKW | 178570 | 1% | 100% | 1 |
| 10 | ANIMAL CELL SUBSTRATE | authKW | 178570 | 1% | 100% | 1 |
Web of Science journal categories |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | BIOLOGY, MISCELLANEOUS | 14018 | 6% | 1% | 11 |
| 2 | Biochemical Research Methods | 1458 | 31% | 0% | 53 |
| 3 | Biotechnology & Applied Microbiology | 997 | 33% | 0% | 56 |
| 4 | Pharmacology & Pharmacy | 325 | 27% | 0% | 46 |
| 5 | Virology | 269 | 10% | 0% | 17 |
| 6 | CYTOLOGY & HISTOLOGY | 91 | 1% | 0% | 2 |
| 7 | Biochemistry & Molecular Biology | 87 | 22% | 0% | 38 |
| 8 | Medicine, Research & Experimental | 73 | 9% | 0% | 15 |
| 9 | Chemistry, Analytical | 59 | 9% | 0% | 16 |
| 10 | Biology | 58 | 6% | 0% | 10 |
Address terms |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | ECT ANIM PATHOGEN MICROORGANISMS | 357140 | 1% | 100% | 2 |
| 2 | MERCK THER EUT | 229587 | 2% | 43% | 3 |
| 3 | BIOASSAY METHODS DEV PHARMACEUT DEV MFG SCI | 178570 | 1% | 100% | 1 |
| 4 | BIOL CONTROLS IMMUNOL MED BIOL SAFETY | 178570 | 1% | 100% | 1 |
| 5 | BIOPROC DEV BIOL VACCINES | 178570 | 1% | 100% | 1 |
| 6 | DISCOVERY BIOL SAFETY ASSESSMENT | 178570 | 1% | 100% | 1 |
| 7 | EFFECT ASSESSMENT | 178570 | 1% | 100% | 1 |
| 8 | MERCK S WP53C350 | 178570 | 1% | 100% | 1 |
| 9 | MERCK VACCINOL | 178570 | 1% | 100% | 1 |
| 10 | PROD HEPATITIS B | 178570 | 1% | 100% | 1 |
Journals |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | BIOLOGICALS | 82075 | 15% | 2% | 25 |
| 2 | JOURNAL OF BIOLOGICAL STANDARDIZATION | 52296 | 6% | 3% | 11 |
| 3 | DEVELOPMENTS IN BIOLOGICAL STANDARDIZATION | 19791 | 5% | 1% | 8 |
| 4 | DEVELOPMENTS IN BIOLOGICALS | 5491 | 1% | 2% | 2 |
| 5 | BIOTECHNIQUES | 2938 | 6% | 0% | 10 |
| 6 | PHARMACEUTICAL SCIENCE & TECHNOLOGY TODAY | 2444 | 1% | 1% | 1 |
| 7 | JOURNAL OF HUMAN VIROLOGY | 1858 | 1% | 1% | 1 |
| 8 | BIOPHARM-THE TECHNOLOGY & BUSINESS OF BIOPHARMACEUTICALS | 1784 | 1% | 1% | 1 |
| 9 | JOURNAL OF PARENTERAL SCIENCE AND TECHNOLOGY | 1499 | 1% | 1% | 1 |
| 10 | HUMAN GENE THERAPY METHODS | 1074 | 1% | 1% | 1 |
Author Key Words |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
LCSH search | Wikipedia search |
|---|---|---|---|---|---|---|---|
| 1 | RESIDUAL DNA | 1269830 | 5% | 89% | 8 | Search RESIDUAL+DNA | Search RESIDUAL+DNA |
| 2 | RESIDUAL HOST CELL DNA | 401781 | 2% | 75% | 3 | Search RESIDUAL+HOST+CELL+DNA | Search RESIDUAL+HOST+CELL+DNA |
| 3 | DNA ONCOGENICITY | 357140 | 1% | 100% | 2 | Search DNA+ONCOGENICITY | Search DNA+ONCOGENICITY |
| 4 | HISTORY OF VACCINES | 357140 | 1% | 100% | 2 | Search HISTORY+OF+VACCINES | Search HISTORY+OF+VACCINES |
| 5 | HOST RESIDUAL DNA | 357140 | 1% | 100% | 2 | Search HOST+RESIDUAL+DNA | Search HOST+RESIDUAL+DNA |
| 6 | RESIDUAL HOST DNA | 357140 | 1% | 100% | 2 | Search RESIDUAL+HOST+DNA | Search RESIDUAL+HOST+DNA |
| 7 | 3D VIRUS CULTURE | 178570 | 1% | 100% | 1 | Search 3D+VIRUS+CULTURE | Search 3D+VIRUS+CULTURE |
| 8 | ANIMAL CELL SUBSTRATE | 178570 | 1% | 100% | 1 | Search ANIMAL+CELL+SUBSTRATE | Search ANIMAL+CELL+SUBSTRATE |
| 9 | ASBV VIROID | 178570 | 1% | 100% | 1 | Search ASBV+VIROID | Search ASBV+VIROID |
| 10 | BIOTECHNOLOGY DRUG | 178570 | 1% | 100% | 1 | Search BIOTECHNOLOGY+DRUG | Search BIOTECHNOLOGY+DRUG |
Core articles |
The table includes core articles in the class. The following variables is taken into account for the relevance score of an article in a cluster c: (1) Number of references referring to publications in the class. (2) Share of total number of active references referring to publications in the class. (3) Age of the article. New articles get higher score than old articles. (4) Citation rate, normalized to year. |
| Rank | Reference | # ref. in cl. |
Shr. of ref. in cl. |
Citations |
|---|---|---|---|---|
| 1 | ANDRE, M , REGHIN, S , BOUSSARD, E , LEMPEREUR, L , MAISONNEUVE, S , (2016) UNIVERSAL REAL-TIME PCR ASSAY FOR QUANTITATION AND SIZE EVALUATION OF RESIDUAL CELL DNA IN HUMAN VIRAL VACCINES.BIOLOGICALS. VOL. 44. ISSUE 3. P. 139 -149 | 16 | 38% | 0 |
| 2 | CHANG, JT , CHEN, YC , CHOU, YC , WANG, SR , (2014) QUANTITATIVE DETECTION OF RESIDUAL PORCINE HOST CELL DNA BY REAL-TIME PCR.BIOLOGICALS. VOL. 42. ISSUE 2. P. 74-78 | 10 | 38% | 2 |
| 3 | LOKTEFF, M , KLINGUER-HAMOUR, C , JULIEN, E , PICOT, D , LANNES, L , NGUYEN, T , BONNEFOY, JY , BECK, A , (2001) RESIDUAL DNA QUANTIFICATION IN CLINICAL BATCHES OF BBG2NA, A RECOMBINANT SUBUNIT VACCINE AGAINST HUMAN RESPIRATORY SYNCYTIAL VIRUS.BIOLOGICALS. VOL. 29. ISSUE 2. P. 123-132 | 10 | 53% | 7 |
| 4 | (1998) REQUIREMENTS FOR THE USE OF ANIMAL CELLS AS IN VITRO SUBSTRATES FOR THE PRODUCTION OF BIOLOGICALS.WHO EXPERT COMMITTEE ON BIOLOGICAL STANDARDIZATION - FORTY-SEVENTH REPORT. VOL. 878. ISSUE . P. 19-56 | 8 | 67% | 0 |
| 5 | GRACHEV, V , MAGRATH, D , GRIFFITHS, E , (1998) WHO REQUIREMENTS FOR THE USE OF ANIMAL CELLS AS IN VITRO SUBSTRATES FOR THE PRODUCTION OF BIOLOGICALS - (REQUIREMENTS FOR BIOLOGICAL SUBSTANCES N DEGREES 50).SAFETY OF BIOLOGICAL PRODUCTS PREPARED FROM MAMMALIAN CELL CULTURE. VOL. 93. ISSUE . P. 141 -171 | 8 | 67% | 0 |
| 6 | ASSAF, SLMR , YOKOMIZO, AY , SERAPICOS, ED , GALLINA, NMF , MACHADO, LD , (1997) TUMORIGENICITY TESTING OF VERO CELLS.ARQUIVOS DE BIOLOGIA E TECNOLOGIA. VOL. 40. ISSUE 3. P. 596 -600 | 5 | 100% | 1 |
| 7 | YANG, H , WEI, ZP , SCHENERMAN, M , (2015) A STATISTICAL APPROACH TO DETERMINING CRITICALITY OF RESIDUAL HOST CELL DNA.JOURNAL OF BIOPHARMACEUTICAL STATISTICS. VOL. 25. ISSUE 2. P. 234 -246 | 6 | 50% | 0 |
| 8 | RIGGIN, A , DAVIS, GC , COPMANN, TL , (1996) REASSESSING THE CONTROL OF RESIDUAL DNA IN BIOPHARMACEUTICALS.BIOPHARM-THE TECHNOLOGY & BUSINESS OF BIOPHARMACEUTICALS. VOL. 9. ISSUE 9. P. 36-& | 11 | 39% | 6 |
| 9 | FERRO, S , FABRE, I , CHENIVESSE, X , (2016) OPTIMIZING A METHOD FOR THE QUANTIFICATION BY QUANTITATIVE REAL-TIME POLYMERASE CHAIN REACTION OF HOST CELL DNA IN PLASMID VECTOR BATCHES USED IN HUMAN GENE THERAPY.HUMAN GENE THERAPY METHODS. VOL. 27. ISSUE 4. P. 159 -170 | 6 | 40% | 0 |
| 10 | SHEN, X , CHEN, XM , TABOR, DE , LIU, Y , ALBARGHOUTHI, M , ZHANG, YF , GALINSKI, MS , (2013) SIZE ANALYSIS OF RESIDUAL HOST CELL DNA IN CELL CULTURE-PRODUCED VACCINES BY CAPILLARY GEL ELECTROPHORESIS.BIOLOGICALS. VOL. 41. ISSUE 3. P. 201-208 | 7 | 33% | 6 |
Classes with closest relation at Level 1 |