Class information for: |
Basic class information |
| Class id | #P | Avg. number of references |
Database coverage of references |
|---|---|---|---|
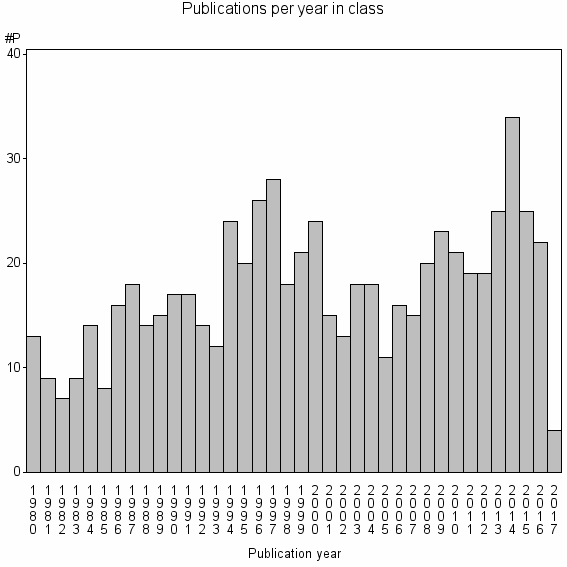
| 16303 | 662 | 39.9 | 27% |
Hierarchy of classes |
The table includes all classes above and classes immediately below the current class. |
| Cluster id | Level | Cluster label | #P |
|---|---|---|---|
| 1 | 4 | ECONOMICS//EDUCATION & EDUCATIONAL RESEARCH//PSYCHOL | 3876184 |
| 320 | 3 | AMERICAN JOURNAL OF PHARMACEUTICAL EDUCATION//MEDICATION ERRORS//AMERICAN JOURNAL OF HOSPITAL PHARMACY | 39312 |
| 1464 | 2 | FOOD AND DRUG LAW JOURNAL//CONFLICT OF INTEREST//ORPHAN DRUGS | 7747 |
| 16303 | 1 | FOOD AND DRUG LAW JOURNAL//MEDICAL DEVICE INNOVATION//DEVICE APPROVAL | 662 |
Terms with highest relevance score |
| rank | Term | termType | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|---|
| 1 | FOOD AND DRUG LAW JOURNAL | journal | 2250093 | 33% | 22% | 220 |
| 2 | MEDICAL DEVICE INNOVATION | authKW | 138374 | 0% | 100% | 3 |
| 3 | DEVICE APPROVAL | authKW | 96087 | 1% | 42% | 5 |
| 4 | CYBERSURGERY | authKW | 92249 | 0% | 100% | 2 |
| 5 | HUMANITARIAN DEVICE EXEMPTION | authKW | 92249 | 0% | 100% | 2 |
| 6 | QA COMPLIANCE VALIDAT | address | 92249 | 0% | 100% | 2 |
| 7 | PREMARKET APPROVAL | authKW | 92245 | 1% | 50% | 4 |
| 8 | MEDICAL DEVICE REGULATION | authKW | 73795 | 1% | 40% | 4 |
| 9 | MEDICAL DEVICES | authKW | 63512 | 4% | 5% | 27 |
| 10 | 21ST CENTURY CURES ACT | authKW | 61498 | 0% | 67% | 2 |
Web of Science journal categories |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | Law | 60987 | 56% | 0% | 374 |
| 2 | Nutrition & Dietetics | 10831 | 34% | 0% | 224 |
| 3 | Food Science & Technology | 5902 | 35% | 0% | 229 |
| 4 | Pharmacology & Pharmacy | 2803 | 39% | 0% | 256 |
| 5 | Health Care Sciences & Services | 412 | 6% | 0% | 38 |
| 6 | Health Policy & Services | 209 | 3% | 0% | 21 |
| 7 | Medical Ethics | 133 | 1% | 0% | 6 |
| 8 | Medicine, General & Internal | 114 | 8% | 0% | 51 |
| 9 | Social Sciences, Biomedical | 102 | 2% | 0% | 12 |
| 10 | Cardiac & Cardiovascular System | 73 | 5% | 0% | 36 |
Address terms |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | QA COMPLIANCE VALIDAT | 92249 | 0% | 100% | 2 |
| 2 | AQ COMPLIANCE VALIDAT | 46125 | 0% | 100% | 1 |
| 3 | CARDIOVASC P NEUROL DEVICESOFF DEV EVAL | 46125 | 0% | 100% | 1 |
| 4 | CART COORDINATING | 46125 | 0% | 100% | 1 |
| 5 | CHIEF SECT PEDIAT CARDIOL | 46125 | 0% | 100% | 1 |
| 6 | CIRCULATORY SUPPORT PROSTHET GRP | 46125 | 0% | 100% | 1 |
| 7 | CIRCULATORY SUPPORT PROTHET GRP | 46125 | 0% | 100% | 1 |
| 8 | CLIN TRANSLAT INFORMAT | 46125 | 0% | 100% | 1 |
| 9 | COMPLIANCE POLICY | 46125 | 0% | 100% | 1 |
| 10 | COURSE FOOD DRUG LAW | 46125 | 0% | 100% | 1 |
Journals |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | FOOD AND DRUG LAW JOURNAL | 2250093 | 33% | 22% | 220 |
| 2 | BIOPHARM-THE APPLIED TECHNOLOGIES OF BIOPHARMACEUTICAL DEVELOPMENT | 16663 | 1% | 5% | 8 |
| 3 | ADMINISTRATIVE LAW REVIEW | 8774 | 2% | 2% | 12 |
| 4 | AMERICAN CRIMINAL LAW REVIEW | 8102 | 2% | 1% | 14 |
| 5 | BIOTECHNOLOGY LAW REPORT | 5047 | 1% | 3% | 4 |
| 6 | AMERICAN JOURNAL OF LAW & MEDICINE | 5030 | 1% | 1% | 8 |
| 7 | JOURNAL OF LEGAL MEDICINE | 4267 | 1% | 1% | 7 |
| 8 | GEORGETOWN LAW JOURNAL | 2285 | 1% | 1% | 8 |
| 9 | TORT & INSURANCE LAW JOURNAL | 2167 | 0% | 2% | 2 |
| 10 | BIOPHARM INTERNATIONAL-THE APPLIED TECHNOLOGIES OF BIOPHARMACEUTICAL DEVELOPMENT | 1706 | 0% | 4% | 1 |
Author Key Words |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
LCSH search | Wikipedia search |
|---|---|---|---|---|---|---|---|
| 1 | MEDICAL DEVICE INNOVATION | 138374 | 0% | 100% | 3 | Search MEDICAL+DEVICE+INNOVATION | Search MEDICAL+DEVICE+INNOVATION |
| 2 | DEVICE APPROVAL | 96087 | 1% | 42% | 5 | Search DEVICE+APPROVAL | Search DEVICE+APPROVAL |
| 3 | CYBERSURGERY | 92249 | 0% | 100% | 2 | Search CYBERSURGERY | Search CYBERSURGERY |
| 4 | HUMANITARIAN DEVICE EXEMPTION | 92249 | 0% | 100% | 2 | Search HUMANITARIAN+DEVICE+EXEMPTION | Search HUMANITARIAN+DEVICE+EXEMPTION |
| 5 | PREMARKET APPROVAL | 92245 | 1% | 50% | 4 | Search PREMARKET+APPROVAL | Search PREMARKET+APPROVAL |
| 6 | MEDICAL DEVICE REGULATION | 73795 | 1% | 40% | 4 | Search MEDICAL+DEVICE+REGULATION | Search MEDICAL+DEVICE+REGULATION |
| 7 | MEDICAL DEVICES | 63512 | 4% | 5% | 27 | Search MEDICAL+DEVICES | Search MEDICAL+DEVICES |
| 8 | 21ST CENTURY CURES ACT | 61498 | 0% | 67% | 2 | Search 21ST+CENTURY+CURES+ACT | Search 21ST+CENTURY+CURES+ACT |
| 9 | CDRH | 61498 | 0% | 67% | 2 | Search CDRH | Search CDRH |
| 10 | NOTIFIED BODIES | 61498 | 0% | 67% | 2 | Search NOTIFIED+BODIES | Search NOTIFIED+BODIES |
Core articles |
The table includes core articles in the class. The following variables is taken into account for the relevance score of an article in a cluster c: (1) Number of references referring to publications in the class. (2) Share of total number of active references referring to publications in the class. (3) Age of the article. New articles get higher score than old articles. (4) Citation rate, normalized to year. |
| Rank | Reference | # ref. in cl. |
Shr. of ref. in cl. |
Citations |
|---|---|---|---|---|
| 1 | HWANG, TJ , SOKOLOV, E , FRANKLIN, JM , KESSELHEIM, AS , (2016) COMPARISON OF RATES OF SAFETY ISSUES AND REPORTING OF TRIAL OUTCOMES FOR MEDICAL DEVICES APPROVED IN THE EUROPEAN UNION AND UNITED STATES: COHORT STUDY.BMJ-BRITISH MEDICAL JOURNAL. VOL. 353. ISSUE . P. - | 17 | 55% | 1 |
| 2 | GREEN, MD , SCHULTZ, WB , (2000) TORT LAW DEFERENCE TO FDA REGULATION OF MEDICAL DEVICES.GEORGETOWN LAW JOURNAL. VOL. 88. ISSUE 7. P. 2119-2145 | 22 | 67% | 14 |
| 3 | ZELENAY, JL , (2005) THE PRESCRIPTION DRUG USER FEE ACT: IS A FASTER FOOD AND DRUG ADMINISTRATION ALWAYS A BETTER FOOD AND DRUG ADMINISTRATION?.FOOD AND DRUG LAW JOURNAL. VOL. 60. ISSUE 2. P. 261-338 | 17 | 74% | 12 |
| 4 | WALSH, CJ , PYRICH, A , (1996) RATIONALIZING THE REGULATION OF PRESCRIPTION DRUGS AND MEDICAL DEVICES: PERSPECTIVES ON PRIVATE CERTIFICATION AND TORT REFORM.RUTGERS LAW REVIEW. VOL. 48. ISSUE 3. P. 883-1041 | 27 | 61% | 17 |
| 5 | KRAMER, DB , XU, S , KESSELHEIM, AS , (2012) HOW DOES MEDICAL DEVICE REGULATION PERFORM IN THE UNITED STATES AND THE EUROPEAN UNION? A SYSTEMATIC REVIEW.PLOS MEDICINE. VOL. 9. ISSUE 7. P. - | 9 | 82% | 20 |
| 6 | MAISEL, WH , (2004) MEDICAL DEVICE REGULATION: AN INTRODUCTION FOR THE PRACTICING PHYSICIAN.ANNALS OF INTERNAL MEDICINE. VOL. 140. ISSUE 4. P. 296-302 | 11 | 79% | 76 |
| 7 | KESSLER, DA , PAPE, SM , SUNDWALL, DN , (1987) THE FEDERAL-REGULATION OF MEDICAL DEVICES.NEW ENGLAND JOURNAL OF MEDICINE. VOL. 317. ISSUE 6. P. 357-366 | 19 | 90% | 41 |
| 8 | SAMUEL, AM , RATHI, VK , GRAUER, JN , ROSS, JS , (2016) HOW DO ORTHOPAEDIC DEVICES CHANGE AFTER THEIR INITIAL FDA PREMARKET APPROVAL?.CLINICAL ORTHOPAEDICS AND RELATED RESEARCH. VOL. 474. ISSUE 4. P. 1053 -1068 | 8 | 80% | 0 |
| 9 | GILMAN, TK , MCCORMICK, BR , (2001) FEDERAL FOOD AND DRUG ACT VIOLATIONS.AMERICAN CRIMINAL LAW REVIEW. VOL. 38. ISSUE 3. P. 819-842 | 10 | 91% | 0 |
| 10 | FREITAG, N , (2004) FEDERAL FOOD AND DRUG ACT VIOLATIONS.AMERICAN CRIMINAL LAW REVIEW. VOL. 41. ISSUE 2. P. 647-669 | 9 | 90% | 2 |
Classes with closest relation at Level 1 |