Class information for: |
Basic class information |
| Class id | #P | Avg. number of references |
Database coverage of references |
|---|---|---|---|
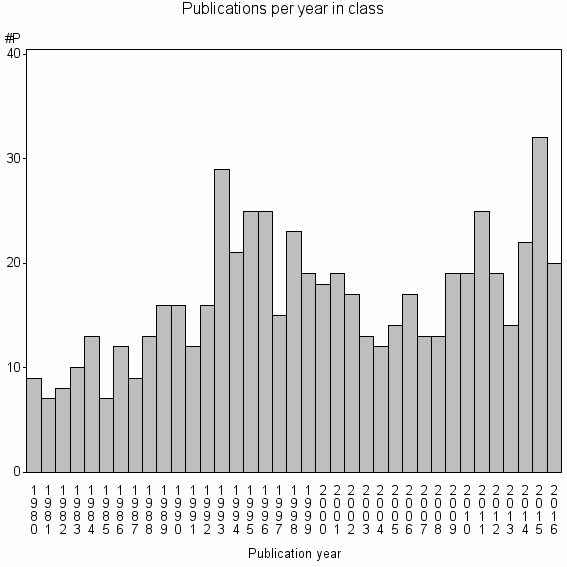
| 17208 | 611 | 42.0 | 72% |
Hierarchy of classes |
The table includes all classes above and classes immediately below the current class. |
| Cluster id | Level | Cluster label | #P |
|---|---|---|---|
| 0 | 4 | BIOCHEMISTRY & MOLECULAR BIOLOGY//CELL BIOLOGY//ONCOLOGY | 4064930 |
| 10 | 3 | IMMUNOLOGY//JOURNAL OF IMMUNOLOGY//IMMUNOL | 134299 |
| 140 | 2 | IMMUNOTHERAPY//DENDRITIC CELLS//CANCER IMMUNOLOGY IMMUNOTHERAPY | 23462 |
| 17208 | 1 | NEWCASTLE DISEASE VIRUS//ACTIVE SPECIFIC IMMUNOTHERAPY ASI//ACTIVE SPECIFIC IMMUNOTHERAPY | 611 |
Terms with highest relevance score |
| rank | Term | termType | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|---|
| 1 | NEWCASTLE DISEASE VIRUS | authKW | 393246 | 12% | 11% | 73 |
| 2 | ACTIVE SPECIFIC IMMUNOTHERAPY ASI | authKW | 249874 | 1% | 100% | 5 |
| 3 | ACTIVE SPECIFIC IMMUNOTHERAPY | authKW | 246008 | 3% | 31% | 16 |
| 4 | CELLULAR IMMUNOL D010 | address | 199899 | 1% | 100% | 4 |
| 5 | TUMOR CELL VACCINES | authKW | 179904 | 1% | 60% | 6 |
| 6 | VIRAL ONCOLYSATE | authKW | 159918 | 1% | 80% | 4 |
| 7 | AF2240 | authKW | 149924 | 0% | 100% | 3 |
| 8 | MTH 68 H | authKW | 149924 | 0% | 100% | 3 |
| 9 | VACCINIA MELANOMA ONCOLYSATE | authKW | 149924 | 0% | 100% | 3 |
| 10 | CELLULAR IMMUNOL | address | 131867 | 8% | 5% | 51 |
Web of Science journal categories |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | Oncology | 8025 | 59% | 0% | 359 |
| 2 | Medicine, Research & Experimental | 1063 | 16% | 0% | 100 |
| 3 | Immunology | 785 | 18% | 0% | 113 |
| 4 | Virology | 505 | 7% | 0% | 45 |
| 5 | Biotechnology & Applied Microbiology | 113 | 7% | 0% | 44 |
| 6 | Surgery | 55 | 7% | 0% | 45 |
| 7 | Radiology, Nuclear Medicine & Medical Imaging | 31 | 4% | 0% | 25 |
| 8 | Genetics & Heredity | 24 | 4% | 0% | 25 |
| 9 | Pharmacology & Pharmacy | 23 | 6% | 0% | 39 |
| 10 | Dermatology | 17 | 2% | 0% | 11 |
Address terms |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | CELLULAR IMMUNOL D010 | 199899 | 1% | 100% | 4 |
| 2 | CELLULAR IMMUNOL | 131867 | 8% | 5% | 51 |
| 3 | ABT 0710 | 99950 | 0% | 100% | 2 |
| 4 | ABT ZELLULARE IMMUNOL G0100 | 99950 | 0% | 100% | 2 |
| 5 | BIOPHARMACEUT TEACHING | 99950 | 0% | 100% | 2 |
| 6 | ABT ZELLULARE IMMUNOL | 66632 | 0% | 67% | 2 |
| 7 | ABT DERMTOL | 49975 | 0% | 100% | 1 |
| 8 | BIOCHEMCHANOCK VIROL | 49975 | 0% | 100% | 1 |
| 9 | CANC PHYS | 49975 | 0% | 100% | 1 |
| 10 | CIRUGIA GEN MASTOL | 49975 | 0% | 100% | 1 |
Journals |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
|---|---|---|---|---|---|
| 1 | CANCER BIOTHERAPY AND RADIOPHARMACEUTICALS | 21388 | 4% | 2% | 25 |
| 2 | CANCER IMMUNOLOGY IMMUNOTHERAPY | 14890 | 6% | 1% | 36 |
| 3 | INTERNATIONAL JOURNAL OF ONCOLOGY | 9231 | 7% | 0% | 42 |
| 4 | JOURNAL OF BIOLOGICAL RESPONSE MODIFIERS | 3682 | 1% | 1% | 6 |
| 5 | CANCER BIOTHERAPY | 2100 | 0% | 2% | 2 |
| 6 | YAKHTEH | 1384 | 0% | 1% | 2 |
| 7 | CANCER GENE THERAPY | 1238 | 1% | 0% | 7 |
| 8 | CANCER AND METASTASIS REVIEWS | 1018 | 1% | 0% | 5 |
| 9 | SEMINARS IN SURGICAL ONCOLOGY | 922 | 1% | 0% | 4 |
| 10 | TECHNOLOGY IN CANCER RESEARCH & TREATMENT | 787 | 1% | 0% | 4 |
Author Key Words |
| Rank | Term | Chi square | Shr. of publ. in class containing term |
Class's shr. of term's tot. occurrences |
#P with term in class |
LCSH search | Wikipedia search |
|---|---|---|---|---|---|---|---|
| 1 | NEWCASTLE DISEASE VIRUS | 393246 | 12% | 11% | 73 | Search NEWCASTLE+DISEASE+VIRUS | Search NEWCASTLE+DISEASE+VIRUS |
| 2 | ACTIVE SPECIFIC IMMUNOTHERAPY ASI | 249874 | 1% | 100% | 5 | Search ACTIVE+SPECIFIC+IMMUNOTHERAPY+ASI | Search ACTIVE+SPECIFIC+IMMUNOTHERAPY+ASI |
| 3 | ACTIVE SPECIFIC IMMUNOTHERAPY | 246008 | 3% | 31% | 16 | Search ACTIVE+SPECIFIC+IMMUNOTHERAPY | Search ACTIVE+SPECIFIC+IMMUNOTHERAPY |
| 4 | TUMOR CELL VACCINES | 179904 | 1% | 60% | 6 | Search TUMOR+CELL+VACCINES | Search TUMOR+CELL+VACCINES |
| 5 | VIRAL ONCOLYSATE | 159918 | 1% | 80% | 4 | Search VIRAL+ONCOLYSATE | Search VIRAL+ONCOLYSATE |
| 6 | AF2240 | 149924 | 0% | 100% | 3 | Search AF2240 | Search AF2240 |
| 7 | MTH 68 H | 149924 | 0% | 100% | 3 | Search MTH+68+H | Search MTH+68+H |
| 8 | VACCINIA MELANOMA ONCOLYSATE | 149924 | 0% | 100% | 3 | Search VACCINIA+MELANOMA+ONCOLYSATE | Search VACCINIA+MELANOMA+ONCOLYSATE |
| 9 | NEWCASTLE DISEASE VIRUS NDV | 119044 | 1% | 26% | 9 | Search NEWCASTLE+DISEASE+VIRUS+NDV | Search NEWCASTLE+DISEASE+VIRUS+NDV |
| 10 | AUTOLOGOUS TUMOR CELL LINES | 99950 | 0% | 100% | 2 | Search AUTOLOGOUS+TUMOR+CELL+LINES | Search AUTOLOGOUS+TUMOR+CELL+LINES |
Core articles |
The table includes core articles in the class. The following variables is taken into account for the relevance score of an article in a cluster c: (1) Number of references referring to publications in the class. (2) Share of total number of active references referring to publications in the class. (3) Age of the article. New articles get higher score than old articles. (4) Citation rate, normalized to year. |
| Rank | Reference | # ref. in cl. |
Shr. of ref. in cl. |
Citations |
|---|---|---|---|---|
| 1 | ZAMARIN, D , PALESE, P , (2012) ONCOLYTIC NEWCASTLE DISEASE VIRUS FOR CANCER THERAPY: OLD CHALLENGES AND NEW DIRECTIONS.FUTURE MICROBIOLOGY. VOL. 7. ISSUE 3. P. 347 -367 | 86 | 55% | 44 |
| 2 | SCHIRRMACHER, V , (2015) ONCOLYTIC NEWCASTLE DISEASE VIRUS AS A PROSPECTIVE ANTI-CANCER THERAPY. A BIOLOGIC AGENT WITH POTENTIAL TO BREAK THERAPY RESISTANCE.EXPERT OPINION ON BIOLOGICAL THERAPY. VOL. 15. ISSUE 12. P. 1757 -1771 | 70 | 71% | 0 |
| 3 | LAM, HY , YEAP, SK , RASOLI, M , OMAR, AR , YUSOFF, K , SURAINI, AA , ALITHEEN, NB , (2011) SAFETY AND CLINICAL USAGE OF NEWCASTLE DISEASE VIRUS IN CANCER THERAPY.JOURNAL OF BIOMEDICINE AND BIOTECHNOLOGY. VOL. . ISSUE . P. - | 54 | 76% | 18 |
| 4 | SCHIRRMACHER, V , (2005) CLINICAL TRIALS OF ANTITUMOR VACCINATION WITH AN AUTOLOGOUS TUMOR CELL VACCINE MODIFIED BY VIRUS INFECTION: IMPROVEMENT OF PATIENT SURVIVAL BASED ON IMPROVED ANTITUMOR IMMUNE MEMORY.CANCER IMMUNOLOGY IMMUNOTHERAPY. VOL. 54. ISSUE 6. P. 587-598 | 38 | 70% | 60 |
| 5 | BAI, FL , TIAN, H , YU, QZ , REN, GP , LI, DS , (2015) EXPRESSING FOREIGN GENES BY NEWCASTLE DISEASE VIRUS FOR CANCER THERAPY.MOLECULAR BIOLOGY. VOL. 49. ISSUE 2. P. 171 -178 | 29 | 63% | 1 |
| 6 | ZHANG, CX , YE, LW , LIU, Y , XU, XY , LI, DR , YANG, YQ , SUN, LL , YUAN, J , (2015) ANTINEOPLASTIC ACTIVITY OF NEWCASTLE DISEASE VIRUS STRAIN D90 IN ORAL SQUAMOUS CELL CARCINOMA.TUMOR BIOLOGY. VOL. 36. ISSUE 9. P. 7121 -7131 | 26 | 70% | 1 |
| 7 | SCHIRRMACHER, V , GRIESBACH, A , AHLERT, T , (2001) ANTITUMOR EFFECTS OF NEWCASTLE DISEASE VIRUS IN VIVO: LOCAL VERSUS SYSTEMIC EFFECTS.INTERNATIONAL JOURNAL OF ONCOLOGY. VOL. 18. ISSUE 5. P. 945 -952 | 25 | 96% | 40 |
| 8 | ZHAO, LX , LIU, HY , (2012) NEWCASTLE DISEASE VIRUS: A PROMISING AGENT FOR TUMOUR IMMUNOTHERAPY.CLINICAL AND EXPERIMENTAL PHARMACOLOGY AND PHYSIOLOGY. VOL. 39. ISSUE 8. P. 725-730 | 30 | 61% | 4 |
| 9 | FU, F , ZHAO, M , YANG, YJ , TONG, GZ , YANG, BF , SONG, C , LI, X , (2011) ANTIPROLIFERATIVE EFFECT OF NEWCASTLE DISEASE VIRUS STRAIN D90 ON HUMAN LUNG CANCER CELL LINE A549.ONCOLOGY RESEARCH. VOL. 19. ISSUE 7. P. 323-333 | 19 | 95% | 4 |
| 10 | SCHIRRMACHER, V , HAAS, C , BONIFER, R , AHLERT, T , GERHARDS, R , ERTEL, C , (1999) HUMAN TUMOR CELL MODIFICATION BY VIRUS INFECTION: AN EFFICIENT AND SAFE WAY TO PRODUCE CANCER VACCINE WITH PLEIOTROPIC IMMUNE STIMULATORY PROPERTIES WHEN USING NEWCASTLE DISEASE VIRUS.GENE THERAPY. VOL. 6. ISSUE 1. P. 63 -73 | 25 | 86% | 79 |
Classes with closest relation at Level 1 |