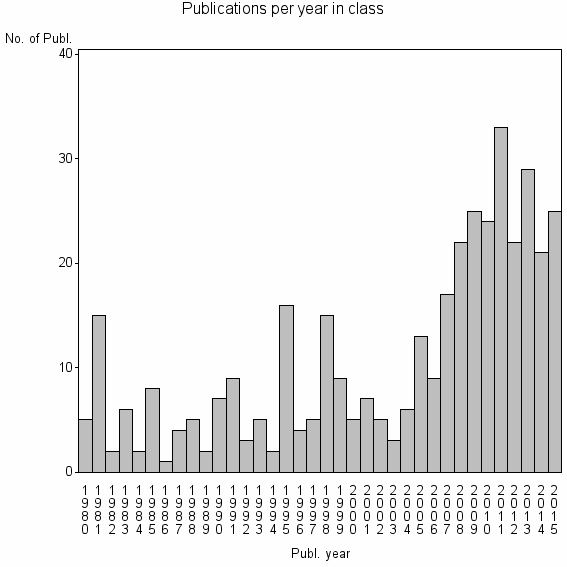
Class information for: |
Basic class information |
| ID | Publications | Average number of references |
Avg. shr. active ref. in WoS |
|---|---|---|---|
| 20633 | 391 | 34.2 | 58% |
Classes in level above (level 2) |
Terms with highest relevance score |
| Rank | Term | Type of term | Relevance score (tfidf) |
Class's shr. of term's tot. occurrences |
Shr. of publ. in class containing term |
Num. of publ. in class |
|---|---|---|---|---|---|---|
| 1 | CHAIR BIOPROC ENGN | Address | 8 | 29% | 6% | 23 |
| 2 | VACCINE PRODUCTION | Author keyword | 6 | 26% | 5% | 19 |
| 3 | RESIDUAL DNA | Author keyword | 5 | 63% | 1% | 5 |
| 4 | SULFATED CELLULOSE | Author keyword | 4 | 75% | 1% | 3 |
| 5 | LEHRSTUHL BIOPROZESSTECH | Address | 3 | 43% | 2% | 6 |
| 6 | CIM MONOLITHIC SUPPORT | Author keyword | 3 | 100% | 1% | 3 |
| 7 | VACCINE PURIFICATION | Author keyword | 3 | 50% | 1% | 4 |
| 8 | EUONYMUS EUROPAEUS LECTIN | Author keyword | 3 | 60% | 1% | 3 |
| 9 | INFLUENZA VACCINE PRODUCTION | Author keyword | 3 | 60% | 1% | 3 |
| 10 | CONTINUOUS CELL LINE | Author keyword | 3 | 42% | 1% | 5 |
Web of Science journal categories |
Author Key Words |
| Rank | Web of Science journal category | Relevance score (tfidf) |
Class's shr. of term's tot. occurrences |
Shr. of publ. in class containing term |
Num. of publ. in class |
LCSH search | Wikipedia search |
|---|---|---|---|---|---|---|---|
| 1 | VACCINE PRODUCTION | 6 | 26% | 5% | 19 | Search VACCINE+PRODUCTION | Search VACCINE+PRODUCTION |
| 2 | RESIDUAL DNA | 5 | 63% | 1% | 5 | Search RESIDUAL+DNA | Search RESIDUAL+DNA |
| 3 | SULFATED CELLULOSE | 4 | 75% | 1% | 3 | Search SULFATED+CELLULOSE | Search SULFATED+CELLULOSE |
| 4 | CIM MONOLITHIC SUPPORT | 3 | 100% | 1% | 3 | Search CIM+MONOLITHIC+SUPPORT | Search CIM+MONOLITHIC+SUPPORT |
| 5 | VACCINE PURIFICATION | 3 | 50% | 1% | 4 | Search VACCINE+PURIFICATION | Search VACCINE+PURIFICATION |
| 6 | EUONYMUS EUROPAEUS LECTIN | 3 | 60% | 1% | 3 | Search EUONYMUS+EUROPAEUS+LECTIN | Search EUONYMUS+EUROPAEUS+LECTIN |
| 7 | INFLUENZA VACCINE PRODUCTION | 3 | 60% | 1% | 3 | Search INFLUENZA+VACCINE+PRODUCTION | Search INFLUENZA+VACCINE+PRODUCTION |
| 8 | CONTINUOUS CELL LINE | 3 | 42% | 1% | 5 | Search CONTINUOUS+CELL+LINE | Search CONTINUOUS+CELL+LINE |
| 9 | HUMAN INFLUENZA A VIRUS | 2 | 38% | 1% | 5 | Search HUMAN+INFLUENZA+A+VIRUS | Search HUMAN+INFLUENZA+A+VIRUS |
| 10 | AGE1CRPIX | 2 | 67% | 1% | 2 | Search AGE1CRPIX | Search AGE1CRPIX |
Key Words Plus |
| Rank | Web of Science journal category | Relevance score (tfidf) |
Class's shr. of term's tot. occurrences |
Shr. of publ. in class containing term |
Num. of publ. in class |
|---|---|---|---|---|---|
| 1 | SCALE MICROCARRIER CULTURE | 23 | 72% | 5% | 18 |
| 2 | ADVENTITIOUS AGENTS | 12 | 86% | 2% | 6 |
| 3 | VERO | 9 | 40% | 4% | 17 |
| 4 | VACCINE PRODUCTION | 4 | 23% | 4% | 14 |
| 5 | ACTIVATED H RAS | 3 | 60% | 1% | 3 |
| 6 | SEED VIRUSES | 3 | 60% | 1% | 3 |
| 7 | ESTABLISHED LINE | 2 | 36% | 1% | 5 |
| 8 | HOST CELL DNA | 2 | 67% | 1% | 2 |
| 9 | GRADE PLASMID DNA | 2 | 43% | 1% | 3 |
| 10 | EMBRYONATED CHICKEN EGGS | 2 | 24% | 2% | 6 |
Journals |
Reviews |
| Title | Publ. year | Cit. | Active references | % act. ref. to same field |
|---|---|---|---|---|
| Designing cell lines for viral vaccine production: Where do we stand? | 2015 | 1 | 64 | 27% |
| The future of cell culture-based influenza vaccine production | 2011 | 16 | 59 | 44% |
| Cell culture-derived influenza vaccines from Vero cells: a new horizon for vaccine production | 2012 | 12 | 48 | 35% |
| Continuous cell lines as a production system for influenza vaccines | 2009 | 45 | 66 | 41% |
| Development and strategies of cell-culture technology for influenza vaccine | 2011 | 9 | 70 | 24% |
| A New Method for the Evaluation of Vaccine Safety Based on Comprehensive Gene Expression Analysis | 2010 | 2 | 16 | 44% |
| Opinion on adventitious agents testing for vaccines: Why do we worry so much about adventitious agents in vaccines? | 2013 | 2 | 8 | 25% |
| Regulatory, biosafety and safety challenges for novel cells as substrates for human vaccines | 2012 | 18 | 50 | 10% |
| Cell culture-based influenza vaccines: A necessary and indispensable investment for the future | 2015 | 0 | 149 | 36% |
| Downstream processing: From egg to cell culture-derived influenza virus particles | 2008 | 3 | 54 | 33% |
Address terms |
| Rank | Address term | Relevance score (tfidf) |
Class's shr. of term's tot. occurrences |
Shr. of publ. in class containing term |
Num. of publ. in class |
|---|---|---|---|---|---|
| 1 | CHAIR BIOPROC ENGN | 8 | 29% | 5.9% | 23 |
| 2 | LEHRSTUHL BIOPROZESSTECH | 3 | 43% | 1.5% | 6 |
| 3 | FAST TRAK SERV EUROPE | 2 | 67% | 0.5% | 2 |
| 4 | MERCK THER EUT | 2 | 43% | 0.8% | 3 |
| 5 | GENOME VALLEY | 1 | 33% | 0.5% | 2 |
| 6 | ANIM NUTR REPROD | 1 | 50% | 0.3% | 1 |
| 7 | BAXTER HYLAND IMMUNO | 1 | 50% | 0.3% | 1 |
| 8 | BIOPHARMACEUT SERV | 1 | 50% | 0.3% | 1 |
| 9 | C T USPHS | 1 | 50% | 0.3% | 1 |
| 10 | DISCOVERY NORTH AMER | 1 | 50% | 0.3% | 1 |
Related classes at same level (level 1) |
| Rank | Relatedness score | Related classes |
|---|---|---|
| 1 | 0.0000139675 | AS03//UNIVERSAL VACCINE//HETEROSUBTYPIC IMMUNITY |
| 2 | 0.0000122334 | HEMAGGLUTININ//ANTIGENIC DRIFT//D222G |
| 3 | 0.0000121017 | RT ASSAY//VIRUS FILTRATION//VIRAL CLEARANCE |
| 4 | 0.0000114576 | POLYMETHYL VINYL ETHER MALEIC ANHYDRATE//POLYMETHYL VINYL ETHER MALEIC ANHYDRIDE//ANIONIC POLYMER |
| 5 | 0.0000113087 | C18L GENE//GO GENE ONTOLOGY//KEGG KYOTO ENCYCLOPEDIA OF GENES AND GENOMES |
| 6 | 0.0000090958 | LIVE ATTENUATED INFLUENZA VACCINE//LAIV//OCULO RESPIRATORY SYNDROME |
| 7 | 0.0000081089 | PB1 F2//INFLUENZA VIRUS//INFLUENZA A VIRUS |
| 8 | 0.0000070558 | HYBRIDOMA//MAMMALIAN CELL CULTURE//CYTOTECHNOLOGY |
| 9 | 0.0000059162 | PSEUDOTYPE BACULOVIRUS//BACMAM//BACULOVIRUS DISPLAY |
| 10 | 0.0000058079 | MOL MED GENE THER Y UNIT//GUTLESS//HELPER DEPENDENT ADENOVIRUS |