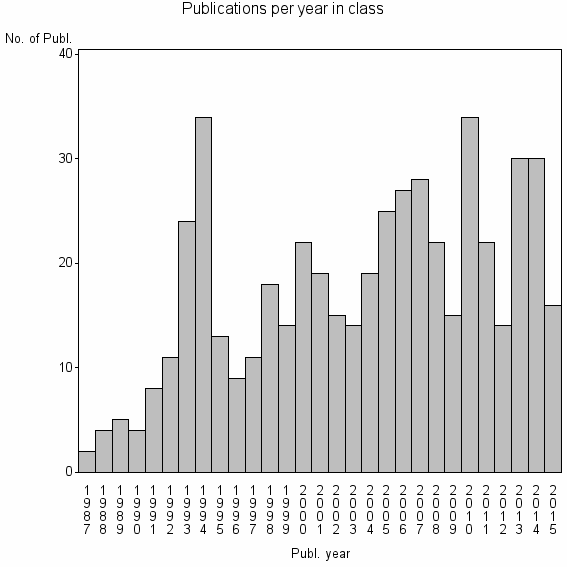
Class information for: |
Basic class information |
| ID | Publications | Average number of references |
Avg. shr. active ref. in WoS |
|---|---|---|---|
| 17998 | 509 | 33.5 | 64% |
Classes in level above (level 2) |
| ID, lev. above |
Publications | Label for level above |
|---|---|---|
| 991 | 9999 | INFORMED CONSENT//RESEARCH ETHICS//MEDICAL ETHICS |
Terms with highest relevance score |
| Rank | Term | Type of term | Relevance score (tfidf) |
Class's shr. of term's tot. occurrences |
Shr. of publ. in class containing term |
Num. of publ. in class |
|---|---|---|---|---|---|---|
| 1 | HIV VACCINE TRIALS | Author keyword | 29 | 63% | 6% | 30 |
| 2 | HIV VACCINES | Author keyword | 17 | 38% | 7% | 36 |
| 3 | WILLINGNESS TO PARTICIPATE | Author keyword | 14 | 48% | 4% | 22 |
| 4 | HIV AIDS VACCINES ETH GRP | Address | 11 | 69% | 2% | 9 |
| 5 | HIV VACCINE | Author keyword | 9 | 18% | 8% | 43 |
| 6 | VACCINE TRIALS | Author keyword | 8 | 34% | 4% | 20 |
| 7 | UNAIDS HIV VACCINE INITIAT | Address | 8 | 75% | 1% | 6 |
| 8 | AIDS VACCINES | Author keyword | 7 | 37% | 3% | 15 |
| 9 | HIV VACCINE PREPAREDNESS | Author keyword | 6 | 100% | 1% | 4 |
| 10 | VACCINE PREPAREDNESS | Author keyword | 4 | 75% | 1% | 3 |
Web of Science journal categories |
Author Key Words |
| Rank | Web of Science journal category | Relevance score (tfidf) |
Class's shr. of term's tot. occurrences |
Shr. of publ. in class containing term |
Num. of publ. in class |
LCSH search | Wikipedia search |
|---|---|---|---|---|---|---|---|
| 1 | HIV VACCINE TRIALS | 29 | 63% | 6% | 30 | Search HIV+VACCINE+TRIALS | Search HIV+VACCINE+TRIALS |
| 2 | HIV VACCINES | 17 | 38% | 7% | 36 | Search HIV+VACCINES | Search HIV+VACCINES |
| 3 | WILLINGNESS TO PARTICIPATE | 14 | 48% | 4% | 22 | Search WILLINGNESS+TO+PARTICIPATE | Search WILLINGNESS+TO+PARTICIPATE |
| 4 | HIV VACCINE | 9 | 18% | 8% | 43 | Search HIV+VACCINE | Search HIV+VACCINE |
| 5 | VACCINE TRIALS | 8 | 34% | 4% | 20 | Search VACCINE+TRIALS | Search VACCINE+TRIALS |
| 6 | AIDS VACCINES | 7 | 37% | 3% | 15 | Search AIDS+VACCINES | Search AIDS+VACCINES |
| 7 | HIV VACCINE PREPAREDNESS | 6 | 100% | 1% | 4 | Search HIV+VACCINE+PREPAREDNESS | Search HIV+VACCINE+PREPAREDNESS |
| 8 | VACCINE PREPAREDNESS | 4 | 75% | 1% | 3 | Search VACCINE+PREPAREDNESS | Search VACCINE+PREPAREDNESS |
| 9 | ACCEPTABILITY OF HEALTHCARE | 4 | 56% | 1% | 5 | Search ACCEPTABILITY+OF+HEALTHCARE | Search ACCEPTABILITY+OF+HEALTHCARE |
| 10 | HIV VACCINE TRIAL | 3 | 40% | 1% | 6 | Search HIV+VACCINE+TRIAL | Search HIV+VACCINE+TRIAL |
Key Words Plus |
| Rank | Web of Science journal category | Relevance score (tfidf) |
Class's shr. of term's tot. occurrences |
Shr. of publ. in class containing term |
Num. of publ. in class |
|---|---|---|---|---|---|
| 1 | EFFICACY TRIALS | 74 | 70% | 12% | 62 |
| 2 | HIGH RISK POPULATIONS | 10 | 29% | 6% | 30 |
| 3 | ECONOMIC COOPERATION | 7 | 67% | 1% | 6 |
| 4 | PROJECT ACHIEVE | 6 | 100% | 1% | 4 |
| 5 | IMMUNODEFICIENCY VIRUS VACCINE | 5 | 40% | 2% | 10 |
| 6 | RISK BEHAVIOR CHANGE | 4 | 67% | 1% | 4 |
| 7 | PARTICIPATE | 4 | 12% | 7% | 34 |
| 8 | VIRUS IMMUNIZATION | 4 | 75% | 1% | 3 |
| 9 | AIDS VACCINE | 4 | 11% | 6% | 33 |
| 10 | PREVENTION TRIALS | 3 | 14% | 5% | 23 |
Journals |
Reviews |
| Title | Publ. year | Cit. | Active references |
% act. ref. to same field |
|---|---|---|---|---|
| HIV vaccine acceptability: a systematic review and meta-analysis | 2010 | 25 | 43 | 79% |
| Barriers of enrolment in HIV vaccine trials: A review of HIV vaccine preparedness studies | 2011 | 7 | 66 | 95% |
| Barriers to participating in an HIV vaccine trial: a systematic review | 2004 | 60 | 33 | 76% |
| Challenges for HIV vaccine dissemination and clinical trial recruitment: If we build it, will they come? | 2004 | 48 | 70 | 54% |
| Barriers to Volunteer Enrollment in HIV Preventive Vaccine Clinical Research Trials: A Review of the Literature | 2011 | 1 | 8 | 88% |
| Socioeconomic status and HIV vaccine preparedness studies in North America | 2015 | 0 | 40 | 90% |
| Motivators to Participation in Actual HIV Vaccine Trials | 2014 | 0 | 22 | 86% |
| Social and behavioral science in HIV vaccine trials: a gap assessment of the literature | 2009 | 3 | 77 | 64% |
| Identifying barriers to participation in prophylactic HIV-1 vaccine trials | 2006 | 0 | 5 | 100% |
| HIV vaccine-induced sero-reactivity: A challenge for trial participants, researchers, and physicians | 2015 | 0 | 14 | 71% |
Address terms |
| Rank | Address term | Relevance score (tfidf) |
Class's shr. of term's tot. occurrences |
Shr. of publ. in class containing term |
Num. of publ. in class |
|---|---|---|---|---|---|
| 1 | HIV AIDS VACCINES ETH GRP | 11 | 69% | 1.8% | 9 |
| 2 | UNAIDS HIV VACCINE INITIAT | 8 | 75% | 1.2% | 6 |
| 3 | VACCINE CLIN BRANCH | 2 | 36% | 1.0% | 5 |
| 4 | HIV AIDS VACCINE ETH GRP | 2 | 67% | 0.4% | 2 |
| 5 | HIV VACCINE SECT | 2 | 67% | 0.4% | 2 |
| 6 | HOSP CLIN IDIB S HIVACAT | 1 | 50% | 0.4% | 2 |
| 7 | S AFRICAN AIDS VACCINE INITIAT | 1 | 100% | 0.4% | 2 |
| 8 | HOPE CLIN | 1 | 19% | 1.2% | 6 |
| 9 | LINKS SOCIAL NETWORK ANAL | 1 | 30% | 0.6% | 3 |
| 10 | HIV VACCINE TRIALS NETWORK | 1 | 14% | 1.2% | 6 |
Related classes at same level (level 1) |
| Rank | Relatedness score | Related classes |
|---|---|---|
| 1 | 0.0000167439 | EQUIPOISE//SHAM SURGERY//DEVELOPING WORLD BIOETHICS |
| 2 | 0.0000126572 | HIV AIDS ABORAT//THAILAND MOPH US CDC ABORAT//AIDS CASES |
| 3 | 0.0000119846 | SIZE OF EPIDEMIC//STOCHASTIC EPIDEMIC MODELS//MULTITYPE EPIDEMIC |
| 4 | 0.0000102797 | REPRINT SERV//HEPATITIS VACCINATION//EDMOND DE ROTH ILD FDN |
| 5 | 0.0000084570 | PATIENT RECRUITMENT//MINORITY RECRUITMENT//TRIAL PARTICIPATION |
| 6 | 0.0000083729 | SERV PSYCHIAT MENTAL HLTH//SYMPTOMATIC VOLUNTEERS//UNDUE INDUCEMENT |
| 7 | 0.0000083124 | PRE EXPOSURE PROPHYLAXIS//POST EXPOSURE PROPHYLAXIS//TREATMENT AS PREVENTION |
| 8 | 0.0000073239 | HGP 30//VACCINE DEV BRANCH//HIV 1 IMMUNOGEN |
| 9 | 0.0000067926 | ASSENT//RESEARCH PARTICIPATION DECISION MAKING//NON THERAPEUTIC RESEARCH |
| 10 | 0.0000067446 | HIV 1 PREVALENCE//HIV PREVALENCE//ETHIONETHERLANDS AIDS PROJECT |