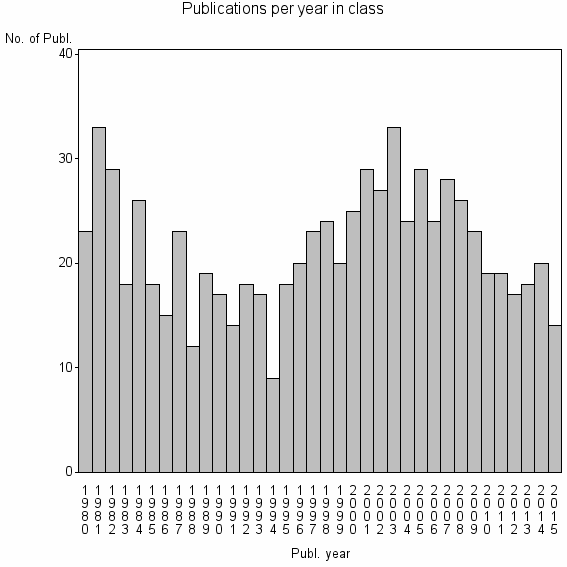
Class information for: |
Basic class information |
| ID | Publications | Average number of references |
Avg. shr. active ref. in WoS |
|---|---|---|---|
| 13458 | 771 | 43.2 | 75% |
Classes in level above (level 2) |
| ID, lev. above |
Publications | Label for level above |
|---|---|---|
| 668 | 12550 | NASOPHARYNGEAL CARCINOMA//EPSTEIN BARR VIRUS//EBV |
Terms with highest relevance score |
| Rank | Term | Type of term | Relevance score (tfidf) |
Class's shr. of term's tot. occurrences |
Shr. of publ. in class containing term |
Num. of publ. in class |
|---|---|---|---|---|---|---|
| 1 | AUSTRALIAN VACCINE DEV TUMOUR IMMUNOL | Address | 4 | 75% | 0% | 3 |
| 2 | EPSTEIN BARR VIRUS NUCLEAR ANTIGEN 1 | Author keyword | 4 | 46% | 1% | 6 |
| 3 | JOINT ONCOL PROGRAM | Address | 4 | 28% | 1% | 11 |
| 4 | CANC UK BIRMINGHAM CANC | Address | 3 | 57% | 1% | 4 |
| 5 | GLY ALA REPEAT | Author keyword | 3 | 57% | 1% | 4 |
| 6 | CLIVE BERGHOFER CANC | Address | 3 | 43% | 1% | 6 |
| 7 | AUSTRALIAN VACCINE DEV | Address | 3 | 24% | 1% | 11 |
| 8 | LATENT MEMBRANE PROTEIN 2A | Author keyword | 2 | 38% | 1% | 5 |
| 9 | LATENT MEMBRANE PROTEIN 2 | Author keyword | 2 | 67% | 0% | 2 |
| 10 | TUMOUR IMMUNOL | Address | 1 | 11% | 2% | 12 |
Web of Science journal categories |
Author Key Words |
Key Words Plus |
| Rank | Web of Science journal category | Relevance score (tfidf) |
Class's shr. of term's tot. occurrences |
Shr. of publ. in class containing term |
Num. of publ. in class |
|---|---|---|---|---|---|
| 1 | NUCLEAR ANTIGEN EBNA1 | 56 | 100% | 2% | 19 |
| 2 | NUCLEAR ANTIGEN 3 | 41 | 85% | 3% | 22 |
| 3 | POSITIVE NASOPHARYNGEAL CARCINOMA | 28 | 81% | 2% | 17 |
| 4 | NUCLEAR ANTIGEN 1 | 14 | 18% | 9% | 68 |
| 5 | LATENT MEMBRANE PROTEIN 2 | 13 | 69% | 1% | 11 |
| 6 | CYCLE ANTIGENS | 12 | 86% | 1% | 6 |
| 7 | PROCESSING FUNCTION | 11 | 100% | 1% | 6 |
| 8 | BURKITTS LYMPHOMA CELLS | 8 | 21% | 5% | 36 |
| 9 | EBV SPECIFIC CD4 | 8 | 100% | 1% | 5 |
| 10 | LYTIC PROTEIN BHRF1 | 8 | 100% | 1% | 5 |
Journals |
Reviews |
| Title | Publ. year | Cit. | Active references | % act. ref. to same field |
|---|---|---|---|---|
| The immunology of Epstein-Barr Virus Induced Disease | 2015 | 1 | 288 | 36% |
| Cellular responses to viral infection in humans: Lessons from Epstein-Barr virus | 2007 | 301 | 140 | 39% |
| Immune defence against EBV and EBV-associated disease | 2011 | 40 | 75 | 45% |
| Human cytotoxic T lymphocyte responses to Epstein-Barr virus infection | 1997 | 497 | 120 | 37% |
| Role of cytotoxic T lymphocytes in Epstein-Barr virus-associated diseases | 2000 | 122 | 79 | 56% |
| The role of virus-specific CD4+T cells in the control of Epstein-Barr virus infection | 2012 | 8 | 34 | 59% |
| The immune response to Epstein-Barr virus | 2004 | 28 | 26 | 81% |
| The interplay between Epstein-Barr virus and the immune system: a rationale for adoptive cell therapy of EBV-related disorders | 2010 | 24 | 105 | 35% |
| Immune escape by Epstein-Barr virus associated malignancies | 2008 | 27 | 86 | 37% |
| Cellular immune controls over Epstein-Barr virus infection: new lessons from the clinic and the laboratory | 2014 | 5 | 92 | 27% |
Address terms |
| Rank | Address term | Relevance score (tfidf) |
Class's shr. of term's tot. occurrences |
Shr. of publ. in class containing term |
Num. of publ. in class |
|---|---|---|---|---|---|
| 1 | AUSTRALIAN VACCINE DEV TUMOUR IMMUNOL | 4 | 75% | 0.4% | 3 |
| 2 | JOINT ONCOL PROGRAM | 4 | 28% | 1.4% | 11 |
| 3 | CANC UK BIRMINGHAM CANC | 3 | 57% | 0.5% | 4 |
| 4 | CLIVE BERGHOFER CANC | 3 | 43% | 0.8% | 6 |
| 5 | AUSTRALIAN VACCINE DEV | 3 | 24% | 1.4% | 11 |
| 6 | TUMOUR IMMUNOL | 1 | 11% | 1.6% | 12 |
| 7 | AUSTRALIAN VACCINE DEV TUMOR IMMUNOL | 1 | 100% | 0.3% | 2 |
| 8 | QIMR IMMUNOTHER Y VACCINE DEV | 1 | 50% | 0.3% | 2 |
| 9 | CLIN COOPERAT GRP | 1 | 13% | 0.9% | 7 |
| 10 | EPSTEIN BARR VIRUS UNIT | 1 | 25% | 0.4% | 3 |
Related classes at same level (level 1) |
| Rank | Relatedness score | Related classes |
|---|---|---|
| 1 | 0.0000250495 | LMP1//LATENT MEMBRANE PROTEIN 1//EBV |
| 2 | 0.0000201170 | SLAM ASSOCIATED PROTEIN SAP//ANTI CMV ANTIBODIES//VARICELLA ARTHRITIS |
| 3 | 0.0000199032 | POST TRANSPLANT LYMPHOPROLIFERATIVE DISORDER//PTLD//POST TRANSPLANT LYMPHOPROLIFERATIVE DISEASE |
| 4 | 0.0000172448 | CMV SPECIFIC T CELLS//ONCOHEMATOL GRP//CYTOMEGALOVIRUS SPECIFIC T CELLS |
| 5 | 0.0000164235 | BZLF1//BRLF1//VIRAL MOL TUMOR DIAGNOST UNIT |
| 6 | 0.0000115091 | MHV 68//MURINE GAMMAHERPESVIRUS//MURINE GAMMAHERPESVIRUS 68 |
| 7 | 0.0000097118 | INFECTIOUS MONONUCLEOSIS//MONONUCLEOSIS//ACUTE EBV INFECTION |
| 8 | 0.0000097100 | XLP//2B4//X LINKED LYMPHOPROLIFERATIVE DISEASE |
| 9 | 0.0000075487 | TAPASIN//TRANSPORTER ASSOCIATED WITH ANTIGEN PROCESSING//PEPTIDE LOADING COMPLEX |
| 10 | 0.0000061500 | CHIMERIC ANTIGEN RECEPTOR//ADOPTIVE CELL THERAPY//CHIMERIC IMMUNE RECEPTOR |